![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
11 Cards in this Set
- Front
- Back
|
what are the types of unit cell? |
1. simple cubic lattice (sc) 2. body-centered cubic lattice (BCC) 3. face-centered cubic lattice (FCC) |
|
|
components of SC (atoms, l, packing eff) |
atoms: 1 l: 2r P.E. : 52.4 |
|
|
components of BCC (atoms, l, packing eff) |
atoms: 2 l: 4r / /3 P.E. : 68 |
|
|
components of FCC (atoms, l, packing eff) |
atoms: 4 l: 2r /2 P.E.: 74 |
|
|
formula for PACKING FACTOR (TETRAGONAL SYMMETRY) |

Back (Definition) |
|
|
formula for DENSITY OF CRYSTALS |

Back (Definition) |
|
|
formula for DENSITY |

Back (Definition) |
|
|
formula for RADIUS |
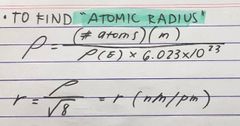
Back (Definition) |
|
|
formula for ENDGE LENGTH OF UNIT CELL |
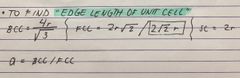
Back (Definition) |
|
|
proof for packing eff of FCC |
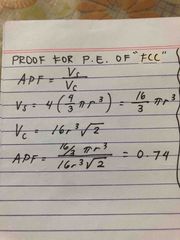
Back (Definition) |
|
|
proof for packing eff of BCC |
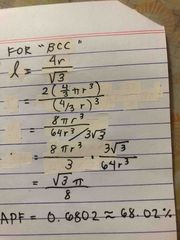
Back (Definition) |

