![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
33 Cards in this Set
- Front
- Back
|
What is distillation |
Distillation is a process used to separate components of a mixture based on their boiling points or by selective boiling and condensation |
|
|
Does distillation always result in complete separation |
It can either result in complete or in partial separation, where the concentration of one species in the mixture is increased |
|
|
When will the mixture boil during distillation |
When the total vapor pressure from contributions of partial reassures of all species equals the external vapour pressure |
|
|
What are 5 types of distillation |
Batch Continuous Fractional Zone Vacuum |
|
|
Describe the process in batch distillation |
Two liquids heated in distillation tower. If A has more volatile components and B has less volatile components then A will be more in vapour phase and B will be retained as a liquid. Equilibrium is reached A is distilled off Remaining compound is enriched in B |
|
|
At least one volatile distillate fraction which had been boiled off had been separately captured as a vapour and condenses to a liquid. |
True |
|
|
Always a residue ( ____ volatile compound not captured as a condensed vapour |
Least |
|
|
How does the composition of the source material, vapours of distilling compound and distillate change during the process |
Changes which time. |
|
|
How does the composition of the source materials, the vapours and the distilling compounds and distillate change in a continuous distillation |
Remains constant |
|
|
How does the composition of the source materials, the vapours and the distilling compounds and distillate change in a continuous distillation - remains constant. How? |
Carefully replenishing the source material and removing fractions from both vapour and liquid phases in system. |
|
|
What does continuous distillation result in |
Better control of the separation process |
|
|
Describe process in continuous distillation |
Liquid mix is continuously fed into the process and separated fractions are constantly removed |
|
|
How many output fractions in continuous distillation |
2 |
|
|
What is vaccum distillation used for |
Compounds with very high BP |
|
|
To boil compounds with very high BP it is better to.. |
Reduce the pressure at which the compounds boil instead of increasing temp. |
|
|
In VD once pressure is lowered to the PvP of the component at a given temp what occurs?what proceeds? |
Boiling. Normal process of distillation |
|
|
Wheree does fractional distillation occur |
In a fractionating column. |
|
|
Describe change in BP Temp in a fractionating column |
Decreasing BP temp as you go higher |
|
|
Describe process in fractional distillation |
Solution is heated. Vapours rise in the column. As the vapours rise it is cooled onto condenser walls or surfaces of the packing material. At the same time it is heated by the vapours beneath it. Each comment with a different BP will condense at different stages into a liquid. |
|
|
Where are the High temp residues in the fractionating column |
At the bottom |
|
|
Where does zone distillation occur |
In a small container |
|
|
The refined matter in ZD is ____ ___ and is in the _____ _____ _____ |
Partially melted Liquid moving zone |
|
|
Condensation of vapour in the ZD IMus in the _____ phase |
Solid |
|
|
Explain process of zone distillation |
Zone heater moves from top to bottom slowly. Condensate with irregular impurity forms and is slowly moved to the bottom of container. When it reaches the bottom all of the impurities are settled and cut so that the most pure part of condensate can be extracted as a product. |
|
|
Can zone distillation be repeated |
Yes. |
|
|
What does repetition result in |
Irregular impurity distribution |
|
|
Graphically representing efficiency of fractional distillation. What are the axis |
X axis is time Y axis is volume change if distillate or the temperature of distillate |
|
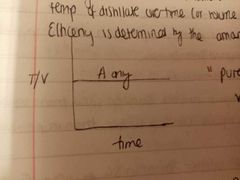
Is this graph representing a good sep or bad SEP why? |
Good. If only pure product is obtained, the BP T will be constant because the temp of the vapours at equilibrium will be constant. Horizontal line = pure product |
|

Good or bad sep? |
Bad. Little to no pure product because temp/volume/Composition of distillate is constantly changing. |
|
Good or bad sep |
?? |
|
|
Where can we apply distillation |
Lab scales Industrial scales Herbal distillate Food processing |
|
|
Is distillation a true SEP method |
Not really. Just used to transfer some volatiles from source material to distillate |
|
|
Examples of distillation |
Distillation of crude oil to get HC Sep of air into its components Distillation of fermentation products |

