![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
90 Cards in this Set
- Front
- Back
|
Structure of Benzene |
Equal angles of 120 Equal C-C bond lengths Equal C-H bond lengths Resonance structures equal |
|
|
Conjugation is _____ stable than isolation. |
More |
|
|
O substitution |
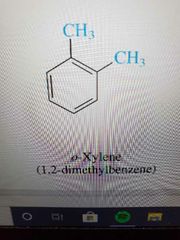
|
|
|
M substituted |
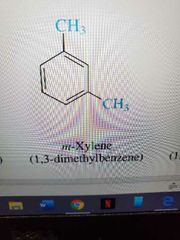
|
|
|
P substituted |
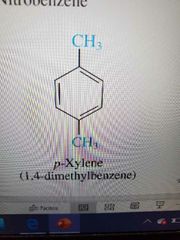
|
|
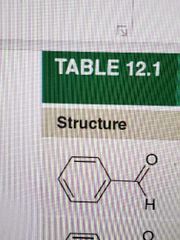
Name |
Benzaldehyde |
|
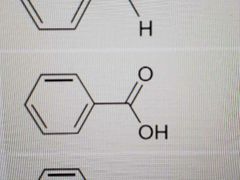
Name |
Benzoic Acid |
|
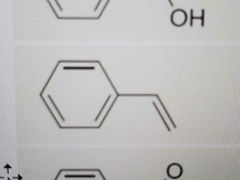
Name |
Styrene |
|
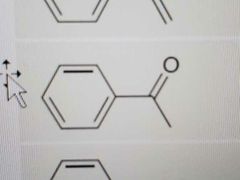
Name |
Acetophenone |
|
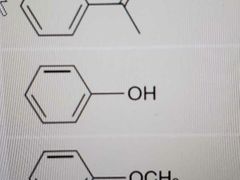
Name |
Phenol |
|
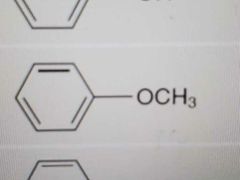
Name |
Anisole |
|
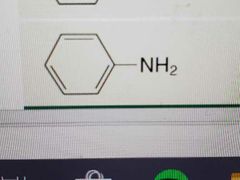
Name |
Aniline |
|
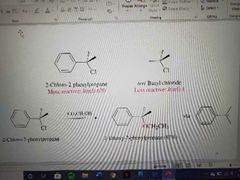
What reaction is shown? |
SN1 of the benzylic halides |
|
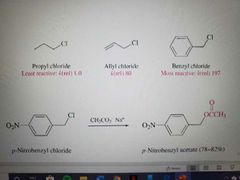
What reaction is shown? |
Sn2 of the Benzylic Halides |
|
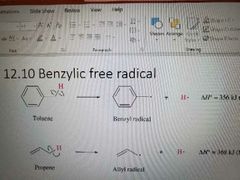
|
Can become radicals like allylic, more stable |
|
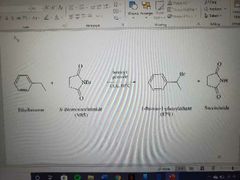
What reaction is shown? |
Benzylic free radical with NBS |
|
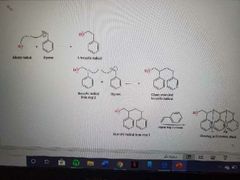
What reaction is shown? |
Polymerization of styrene |
|
|
Huckel's Rule |
Aromatic = (4n+2) = pi electrons Antiaromatic = (4n) = pi electrons |
|
|
Completely conjugated rings with more than 6 carbons. |

Annulenes |
|
|
Can ions be aromatic? |
Yes |
|
|
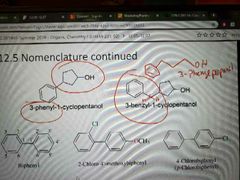
Directly attached, benzene group called "phenyl" |
|
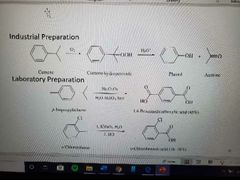
What reactions are shown? |
Oxidation at the Benzylic position |
|
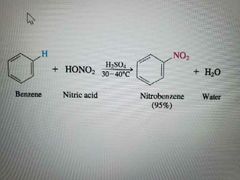
What reaction is shown? |
Nitration |
|
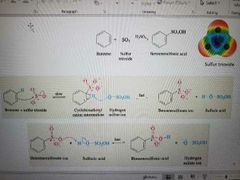
What reaction is shown? |
Sulfonation |
|
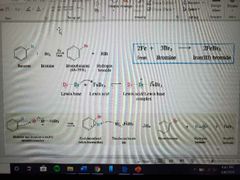
What reaction is shown? |
Halogenation |
|
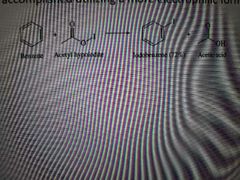
What reaction is shown below? |
Iodination |
|
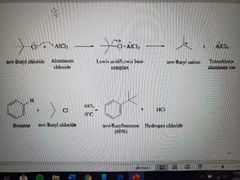
What reaction is shown below? |
Friedel-Crafts Alkylation |
|
|
What reagents are used in Friedel-Crafts Alkylation? |
A carbon chain attached to a halide and AlCl3. |
|
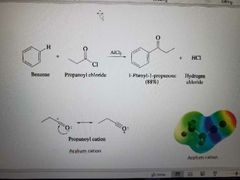
What reaction is shown below? |
Friedel-Crafts Acylation |
|
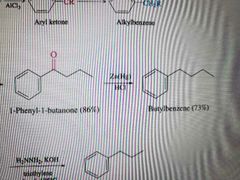
What reaction is shown below? |
Clemmensen reduction |
|
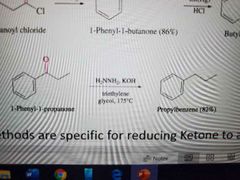
What reaction is shown below? |
Wolff-Kishner reduction |
|
|
What is the rate law of SnAR? |
Second order. Electron withdrawing groups stabilize. Electronegative halides increase the rate of anion formation. |
|
|
Nitrobenzene to aniline reactions. What reactants are used? |
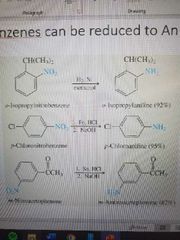
|
|
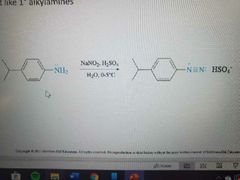
What reaction is shown? |
Nitrosation of Arylamines Only works with 1° amines |
|
|
Reactions with N2 as a psyeudohalide |
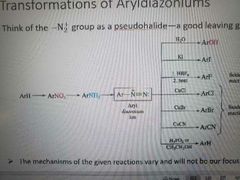
|
|
|
Azo Coupling of Diazonium Salt |
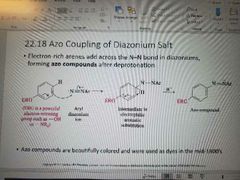
|
|
|
How to name organometallic compounds |
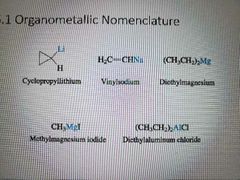
|
|

What reaction is shown? |
Preparation of Organolithium Compounds |
|
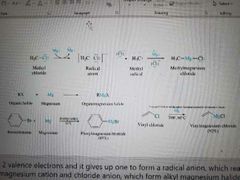
What reaction is shown? |
Preparation of organomagnesium compounds |
|
|
What reagents are used to create organometals? |
Diethyl ether or THF |
|
|
Are organometallic carbons basic or acidic? |
They are Bronsted bases (accept H) |
|
|
What happens when an organometallic and Gringard reagent react with ketones and aldehydes? |
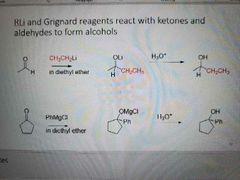
Alcohols are formed |
|
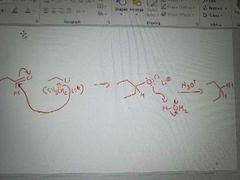
What mechanism is shown? |
Addition of organometallic reagents to ketone or aldehyde (to form alcohols) |
|
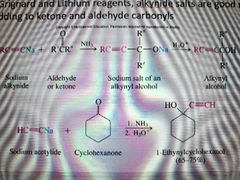
What reaction is shown? |
Creation of acetylenic alcohols |
|
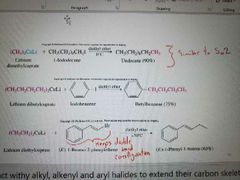
What reaction is shown? |
Organocuprate (Gilman Reagents) reacting with RX and ARX to make C-C bonds Inversion of sterochem Only works with methyl and primary. If secondary or tertiary, E2 occurs |
|
|
What reagents are used for organocuprate reagent reactions? |
Diethyl ether or THF |
|
|
Memorize |

|
|
|
Review of how alcohols are made: |
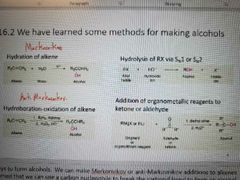
|
|
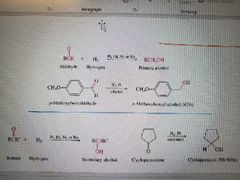
What reaction is shown? |
Metal Catalyzer Hydrogenation of Aldehydes or Ketones |
|
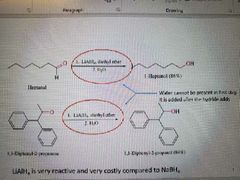
What reaction is shown? |
Lithium aluminum hydride reduction Works with ketones, Aldehydes, and acyl compounds |
|
|
What kind of reagents are used to reduce ketones and aldehydes to alcohols? |
LiAlH4 or NaBH4 and H2O |
|
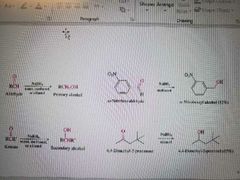
What reactions are shown? |
Sodium Borohydride reduction of Aldehydes or ketones to form alcohols |
|
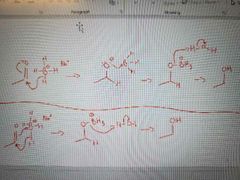
What mechanism is shown? |
Reduction of aldehydes and ketones to form alcohols, with hydride reagents |
|
What mechanism is shown? |
Lithium aluminum hydride reduction of carboxylic acids to primary alcohol |
|
|
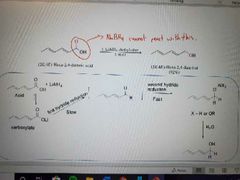
What reagents are used for epoxide ring openings to make alcohols? |
Gringard reagent (organometallic reagents), 1. diethyl ether, 2. H3O+ |
|
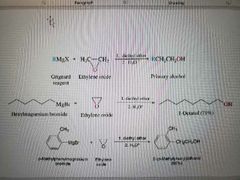
What reaction is shown? |
Formation of alcohols from the ring opening of epoxides using organometals (nucleophiles) |
|
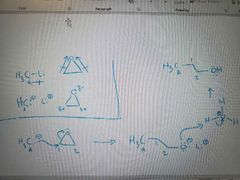
What mechanism is shown? |
Ring opening of an epoxide with an organometallic reagent |
|
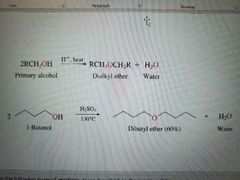
What reaction is shown? |
Acid catalyzed dialkyl ether formation from PRIMARY alcohols |
|
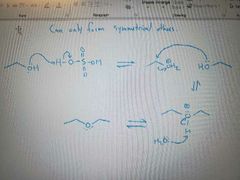
What mechanism is shown? |
Conversion of alcohols to ethers |
|
|
What reagents are needed for Fischer Esterification? |
Alcohol, carboxylic acid, and acid catalyst |
|
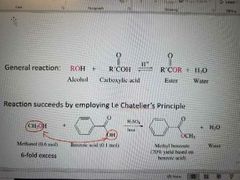
What reaction is shown? |
Fischer Esterification Succeeds by employing Le Chat's. Moves H2O to shift equilibrium to the left. Uses Dean-Stark trap (Benzene/H2O azetrope) |
|
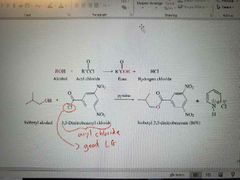
What reaction is shown? |
Creation of esters using reactive acyl halides with alcohol |
|
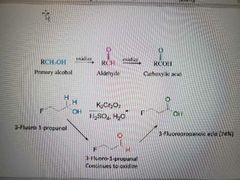
What reaction is shown? |
Oxidation of primary alcohols to carboxylic acids via aldehydes 1° alcohols form carboxy acids 2° alcohols form ketones 3° alcohols do not react |
|
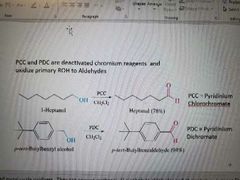
What reaction is shown? |
Oxidation of primary alcohols to aldehydes |
|
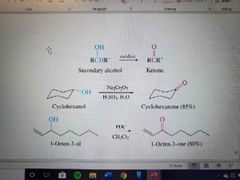
What reaction is shown? |
Oxidation of secondary alcohols to ketones |
|
|
What is the mechanism of the oxidation of secondary alcohols using chromate compounds? |

There is a chromic acid oxidizer which attaches and creates ester. The compound reacts with water and falls apart, which creates oxidized compound (ketone) |
|
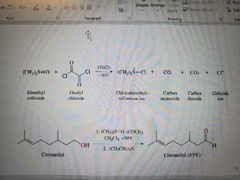
What reaction is shown? |
Oxidation of alcohols using Swern conditions |
|
|
In the body, how are alcohols oxidized? |
NAD+ in alcohol dehydrogenase |
|
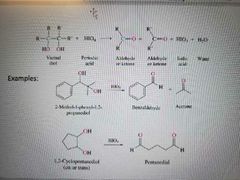
What reaction is shown? |
Oxidative cleavage of vicinal diols using HIO4 |
|
|
How to name thiols: |
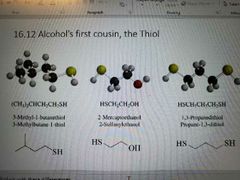
No H bonding in thiols Better nucleophiles Better leaving groups Stronger acids |
|
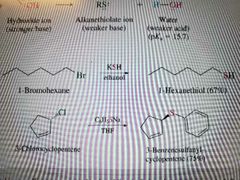
What reaction is shown? |
Sn2 using sulfide or thiols |
|
|
Nomenclature of ethers and epoxides |
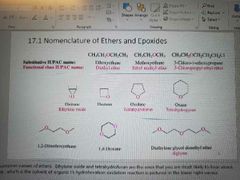
|
|
|
Nomenclature of sulfides |
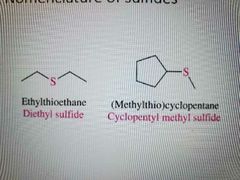
|
|
|
What is a crown ether? |
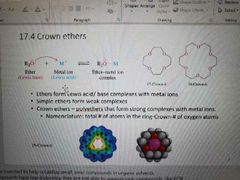
|
|
What reaction is shown? |
Crown ether reaction |
|
|
Reminder of ways to make ethers: |
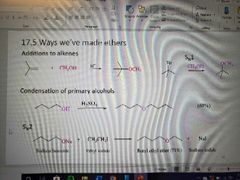
|
|

What reaction is shown? |
Williamson Ether Synthesis |
|
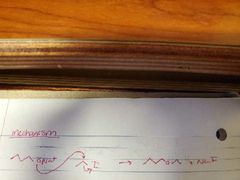
What mechanism is shown? |
Williamson Ether Synthesis |
|
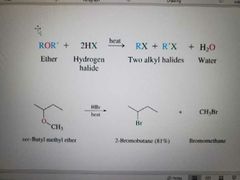
What reaction is shown? |
Acid catalyzed cleavage of ethers to form alkyl halide |
|
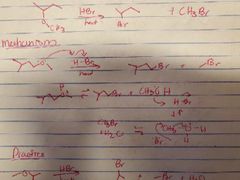
What mechanism is shown? |
Acid catalyzed cleavage of ethers to form alkyl halides |
|
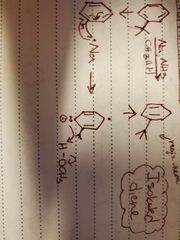
What reaction is shown? |
Birch reduction |
|
|
What reagents are used in the Clemmensen Reduction? |
Zn(Hg) and HCL |
|
|
What reagents are used in the Wolff-Kishner Reduction |
H2NNH2, KOH, triethylene glycol, at 175°C |
|
|
Memorize |
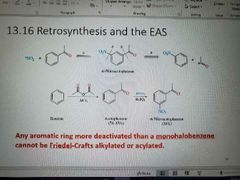
|
|
|
What reagents can be used to oxidize the Benzylic position to carboxylic acids? |
Na2Cr2O7, H2O, H2SO4, heat Or 1. KMnO4, H2O 2. HCl |
|
|
What reagents are used in the Birch Reduction? |
Na, NH3, CH3CH2OH |
|
|
Memorize |
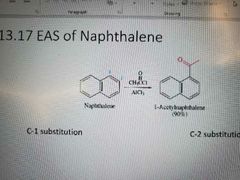
|
|
|
Memorize |

|
|
|
Memorize |
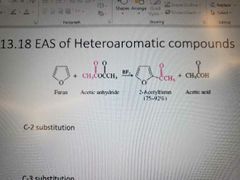
|
|
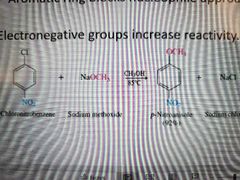
What reaction is shown? |
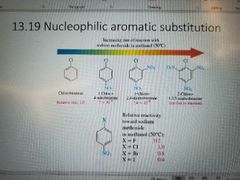
Nucleophilic aromatic substitution (SnAr) Second order |

