![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
26 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
What are the 3 gas laws? |
Boyle's Law Charles' Law Pressure Law |
|
|
|
What does Boyle's Law explain? |
How the temperature and volume of a gas vary when the temperature remains constant |
Isothermal P is inversely proportional to V |
|
|
How do pressure and volume vary for an isothermal reaction (Boyle's Law)? |
The pressure for a fixed mass of gas is inversely proportional to its volume when kept at constant temperature |
|
|
|
Explain Boyle's law |
The pressure of a gas at constant temperature is increased if the volume is reduced because gas molecules travel less distance between impacts due to the reduced volume There are more impacts per second so pressure is increased |
|
|
|
What is a useful equation for Boyle's law? |
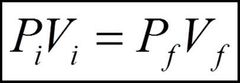
|
P is the pressure acting on a gas of volume V |
|
|
What does the pressure law explain? |
How the temperature and pressure of a gas varies when the volume remains constant |
P is proportional to T |
|
|
Explain the Pressure law |
For a fixed volume of gas increasing the temperature increases the kinetic energies of the particles
Due to the increased kinetic energies of the particles they hit the walls of their container more often causing the pressure to increase |
|
|
|
How do pressure and temperature vary for an isochoric reaction (Pressure Law)? |
The pressure for a fixed mass of gas is directly proportional to the absolute temperature of the gas for a fixed volume |
|
|
|
What is a useful equation for the Pressure law? |
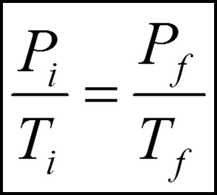
|
|
|
|
How do volume and temperature vary for an isothermic reaction (Charles' Law)? |
At constant pressure the volume of a fixed mass of an ideal gas is directly proportional to its absolute temperature |
|
|
|
What does Charles's law explain? |
How the volume and temperature of a gas vary when the pressure is constant |
Isobaric |
|
|
What is a useful equation for Charles' law? |
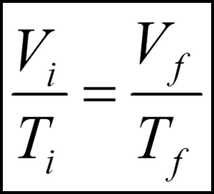
|
|
|
|
Explain Charles's law |
As temperature increases volume must increase as for constant pressure increase temperature = increased kinetic energy So for the number of wall collisions per second (pressure) to remain constant the walls of the container need to move further apart |
|
|
|
What is the equation used to describe the final pressure volume and temperature of a gas after changes have been applied to it? |
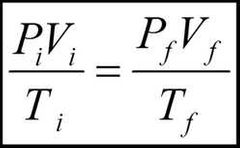
|
|
|
|
What are the 7 assumptions made for an ideal gas? |
There are a very large number of identical, small, hard spherical molecules
The molecules have negligible volume compared to the container
The molecules show random motion (ranges of speeds and directions) with distribution of velocities about a mean velocity
Newtons laws of motion can be applied to the molecules
Collisions are elastic and all motion is frictionless
The time spent between collisions is much larger than the time spent colliding
No intermolecular forces acting between molecules other than when they collide |
|
|
|
What is absolute zero? |
The temperature when all molecular motion ceases and the pressure of a gas drops to zero |
|
|
|
What is the value of absolute zero in Kelvin and degrees Celsius? |
0K -273.15 °C |
|
|
|
What is Avogadro's law? |
Equal volumes of different gases at the same pressure and temperature will have the same number of particles |
|
|
|
What is Avogadro's constant? |
6.02x10^23 mol^-1 |
|
|
|
What equation links number of moles molecular mass and mass of substance? |
n = M/Mr |
|
|
|
What equation links number of moles number of particles and Avogadro's constant? |
n = N/NA |
|
|
|
How was the boltzmann constant calculated? |
Molar gas constant/Avogadro's constant R/NA |
|
|
|
What is the value of the molar gas constant? |
8.31 J K^-1 mol^-1 |
|
|
|
What is the value for the boltzmann constant? |
1.38x10^-23 J K^-1 |
|
|
|
What are the two ideal gas equations? |
pV = nRT pV = NkT |
|
|
|
How do you calculate the work done to move a gas? |
Work Done = Force x Distance p = Force/Area --> Force = Pressure x Area Work Done = Pressure x Area x Volume --> Work Done = Pressure x Change in Volume |
|

