![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
155 Cards in this Set
- Front
- Back
|
where do viroids replicate? Virusoids?
|
- viroids: nucleus
- virusoids: cytoplasm |
|
|
list the building blocks of a non-enveloped virus starting from the smallest part to the largest
|
1. protein subunit
2. structure unit 3. assembly unit 4. capsomer 5. capsid 6. nucleocapsid |
|
|
what are four constituents of the viral envelope?
|
1. peplomer
2. spike proteins 3. matrix proteins 4. lipids |
|
|
where does the enveloped virus acquire its lipid bilayer?
|
form the host cell
|
|
|
what is the function of spike proteins and peplomers?
|
to attach to a specific host cell receptor and bring the virus into the cell
|
|
|
what gives a spike protein or peplomer its host specificity?
|
carbohydrate side chain
|
|
|
which type of viruses are most sensitive to disinfectants and the environment?
|
enveloped viruses
|
|
|
which types of viruses tend to be seasonal?
|
enveloped viruses
|
|
|
explain the basic pathogenesis of how an enveloped virus, after replication spreads the infection to other cells
|
after replication, it buds out of the host cell, causing chronic, persistent infections, and disease
|
|
|
why is an enveloped virus more sensitive to detergent than a non-enveloped virus?
|
because the lipid bilayer is destroyed by detergent and the virus cannot survive without the envelope
|
|
|
why are infections with non-enveloped viruses typically self-limiting, as opposed to enveloped viruses, which are chronic?
|
because they lyse the host cell to reproduce
|
|
|
what are the two types of viral symmetry?
|
helical and icosahedral (cubical)
|
|
|
list the possible types of nucleic acid that a virus could contain in its nucleocapsid
|
- DNA: ss, ds, monopartite, multipartite
- RNA: ss, ds, monopartite, multipartite - retroviruses are diploid; all others are haploid - viruses never have both DNA and RNA |
|
|
list four families of DNA, non-enveloped viruses
|
1. Hepadnaviridae
2. Parvoviridae 3. Papoviridae 4. Adenoviridae |
|
|
list three families of DNA, enveloped viruses
|
1. Herpesviridae
2. Iridoviridae 3. Poxviridae |
|
|
list four families of RNA, non-enveloped viruses
|
1. Picornaviridae
2. Caliciviridae 3. Reoviridae 4. Birnaviridae |
|
|
list nine families of RNA, enveloped viruses
|
1. Togaviridae
2. Coronaviridae 3. Paramyxoviridae 4. Rhabdoviridae 5. Orthomyxoviridae 6. Arenaviridae 7. Bunyaviridae 8. Retroviridae 9. Flaviviridae |
|
|
what is the function of non-structural proteins?
|
- enzymes
- replication - regulatory proteins |
|
|
what is the function of integrase?
|
integrates the proviral DNA of retroviruses into the host genome
|
|
|
what are the suffixes and the correct font style of virus Order, Family, Genus, Species, strain?
|
- Order: -virales, italics
- Family: -viridae, italics - Genus: -virus, italics - species/strain: any name, non-italicized |
|
|
ability to become infected
|
susceptibility
|
|
|
ability to replicate and produce progeny viruses (as allowed by the cell)
|
permissivity
|
|
|
location of first infected cells, and frequently not the site of disease signs
|
portal of entry
|
|
|
infection resulting in infectious progeny
|
productive infection
|
|
|
infection, where replication is initiated, but not completed, has limited gene expression, and produces no infectious progeny
|
abortive infection
|
|
|
infection where permissivity is transient or proportional to the cell population; few virions released
|
restrictive infection
|
|
|
infection where viral genomes are integrated into the cellular genomes, but not expressed; therefore, no infectious progeny
|
latent infection
|
|
|
infection characterized by activation of latency, new infectious progeny, disease flair-up, or a new immune response
|
recrudescence
|
|
|
what is a common reason why a viral infection may be abortive?
|
because it is infecting a susceptible, but not permissive cell
|
|
|
susceptibility
|
ability to become infected
|
|
|
permissivity
|
ability to replicate and produce progeny viruses (as allowed by the cell)
|
|
|
portal of entry
|
location of first infected cells, and frequently not the site of disease signs
|
|
|
productive infection
|
infection resulting in infectious progeny
|
|
|
abortive infection
|
infection, where replication is initiated, but not completed, has limited gene expression, and produces no infectious progeny
|
|
|
restrictive infection
|
infection where permissivity is transient or proportional to the cell population; few virions released
|
|
|
latent infection
|
infection where viral genomes are integrated into the cellular genomes, but not expressed; therefore, no infectious progeny
|
|
|
recrudescence
|
infection characterized by activation of latency, new infectious progeny, disease flair-up, or a new immune response
|
|
|
describe the 10-step virus replication cycle
|
1. attachment
2. penetration 3. uncoating (nucleocapsid disintegrates) 4. transcription of early mRNA 5. translation of early proteins 6. replication of viral DNA 7. transcription of late RNA 8. translation of late proteins 9. assembly of virions 10. release |
|
|
what part of the virus binds to cellular receptors in order to gain entry to the cell?
|
viral attachment proteins
|
|
|
what are the three types of viral penetration, describe the mechanism, and what type of viruses use them.
|
1. translocation: diffusion through the cell membrane; used by non-enveloped viruses
2. endocytosis: entire virus is enveloped into a phagosome and is activated when it becomes a phagolysosome; enveloped viruses use this 3. fusion: lipid envelope becomes part of the cell membrane and the naked nucleocapsid enters the cell; used by some enveloped viruses |
|
|
viral non-structural proteins that are involved with transcription, replication of viral nucleic acid, and cleavage of viral proteins
|
virion-associated enzymes
|
|
|
viral non-structural proteins that are involved with controlling the temporal sequence of viral genome expression (up-regulating and down-regulating cellular genes)
|
regulatory proteins
|
|
|
what type of cellular genes are up-regulated by viral regulatory proteins?
|
Transactivators and oncogene products (such as growth factors, tumor suppressors)
|
|
|
viral non-structural proteins that are involved with the modulation of infection by acting on other uninfected cells be (1) negating the effects of IFN, TNF, etc. and (2) reduce the inflammatory response and complement activation?
|
virokines
|
|
|
in what two basic ways do virokines modulate the infection by acting outside the host cell?
|
1. negate the effects of IFN, TNF, etc.
2. reduce the inflammatory response, complement activation, etc. |
|
|
virion-associated enzymes
|
viral non-structural proteins that are involved with transcription, replication of viral nucleic acid, and cleavage of viral proteins
|
|
|
regulatory proteins
|
viral non-structural proteins that are involved with controlling the temporal sequence of viral genome expression (up-regulating and down-regulating cellular genes)
|
|
|
the process of producing virions after replication, transcription, and translation of viral components.
|
assembly
|
|
|
aligning of virions with the cell membrane before release
|
maturation
|
|
|
assembly
|
the process of producing virions after replication, transcription, and translation of viral components.
|
|
|
maturation
|
aligning of virions with the cell membrane before release
|
|
|
what is the basic process of maturation and release of an enveloped virus?
|
- viral glycoproteins and matrix proteins are put into the host cell membrane
- the viral nucleocapsid aligns with the viral matrix proteins - the virion buds from the cell and is released as a free infectious virion |
|
|
where do most RNA viruses replicate?
|
the cytoplasm
|
|
|
what is the process of (+)-strand RNA virus genome replication
|
- parental (+) strand is used as mRNA for the synthesis of replication proteins
- the (-) strand is made from the (+) strand template - the (+) strand is synthesized into many progeny strands from the (-) strand |
|
|
what is a major difference between the virion of a (+) strand and a (-) strand RNA virion?
|
(-) strand RNA viruses must carry polymerase enzymes in the virion in order to synthesize (+) strand RNA after infection.
|
|
|
what is the process of (-) strand RNA virus genome replication?
|
- (+) strand RNA is produced from the parental (-) strand and the polymerase enzymes carried in the virion
- the resulting (+) strand RNA is used as mRNA to synthesize viral proteins - the (+) strand is also used to synthesize progeny (-) sense progeny strands |
|
|
during the replication cycle, what type of nucleic acid do all viruses share?
|
(+) strand RNA, somewhere in the cycle, which serves as mRNA for the translation of viral proteins
|
|
|
Define the first six Baltimore System classifications of viruses
|
I. dsDNA
II. ssDNA III. dsRNA IV. (+) sense RNA V. (-) sense RNA VI. (+) sense RNA that use reverse transcriptase to make DNA |
|
|
where do DNA viruses replicate? What is a notable exception?
|
they replicate in the nucleus, except for Poxviridae
|
|
|
what do viral early genes and late genes encode for?
|
- early genes: non-structural proteins
- late genes: structural proteins |
|
|
how does size of the DNA virus relate to replication?
|
the smaller DNA viruses are more dependent on the host cell, whereas larger DNA viruses have more control over the replication of their genomes
|
|
|
what is required for DNA polymerase to begin the viral genome replication process in the nucleus?
|
a primer
|
|
|
name four families of dsDNA viruses
|
1. Papoviridae
2. Adenoviridae 3. Herpesviridae 4. Poxviridae |
|
|
why can Poxviridae replicate in the cytoplasm, whereas other dsDNA viruses must use the cell nucleus?
|
because Poxviridae carry all of the components necessary for viral transcription and translation in the virion
|
|
|
name two families of ssDNA viruses
|
1. Parvoviridae
2. Circoviridae |
|
|
what is a dependovirus? Give an example of one.
|
a virus with such a small / incomplete genome, that it needs help from another virus to replicate. Example: Adeno-associated parvoviruses
|
|
|
what is one family of partially-ds gapped DNA viruses?
|
Hepadnaviridae
|
|
|
what steps in viral replication are unique to the Hepadnaviridae (partially-ds gapped DNA viruses)?
|
1. gap repair precedes mRNA synthesis
2. the mRNA encodes reverse transcriptase |
|
|
what two families of viruses use reverse transcriptase?
|
1. Hepadnaviridae
2. Retroviridae |
|
|
what are three reasons that viral replication is error-prone?
|
1. poor fidelity of DNA polymerase
2. rapid rate of replication 3. RNA viruses lack proof-reading mechanisms |
|
|
why do RNA viruses mutate faster than DNA viruses?
|
because RNA is more susceptible to damage and RNA viruses lack the ability to proof-read during replication of their genomes
|
|
|
the "original" virus (e.g. the lab-adapted "parent" strain)
|
wild-type
|
|
|
a virus newly obtained from a host
|
field isolate
|
|
|
different wild-types of the same virus
|
strain
|
|
|
strains that have mutated from the wild-type
|
variant virus strains; variants
|
|
|
wild-type
|
the "original" virus (e.g. the lab-adapted "parent" strain)
|
|
|
field isolate
|
a virus newly obtained from a host
|
|
|
strain
|
different wild-types of the same virus
|
|
|
variant virus strains; variants
|
strains that have mutated from the wild-type
|
|
|
what are the four "types" used to relate strains of viruses?
|
1. serotype: related antigenically
2. genotype: related genetically 3. biotype 4. pathotype: e.g. one causes disease and one does not |
|
|
what are the two types of viral mutations and what is the difference between the two?
|
1. spontaneous mutations: no mutagens involved
2. induced mutations: mutagens involved |
|
|
acquisition or lost ability of a virus to infect a new host; cross-species infection, causing new diseases in a new host
|
host range mutations
|
|
|
the main mechanism by which viruses become resistant to antiviral drugs
|
drug resistance mutation
|
|
|
change of a virus characterized by an increased ability to evade the specific immune system
|
antibody escape mutations
|
|
|
viruses that have mutated to a wild-type phenotype from a modified-live vaccine strain
|
revertants
|
|
|
host range mutations
|
acquisition or lost ability of a virus to infect a new host; cross-species infection, causing new diseases in a new host
|
|
|
antibody escape mutations
|
change of a virus characterized by an increased ability to evade the specific immune system
|
|
|
revertants
|
viruses that have mutated to a wild-type phenotype from a modified-live vaccine strain
|
|
|
mutations in essential genes, resulting in a dead virus
|
lethal mutations
|
|
|
selective removal of a portion of viral genome (gene or fragment) and the function it encodes
|
deletion mutants
|
|
|
viruses with a conditional lethal mutation that allows the virus to grow at elevated or reduced temperatures
|
temperature-sensitive mutants
|
|
|
lethal mutations
|
mutations in essential genes, resulting in a dead virus
|
|
|
deletion mutants
|
selective removal of a portion of viral genome (gene or fragment) and the function it encodes
|
|
|
temperature-sensitive mutants
|
viruses with a conditional lethal mutation that allows the virus to grow at elevated or reduced temperatures
|
|
|
during a mixed infection, the viral genome from one virus is enclosed in the capsid or envelope of another virus
|
phenotypic mixing
|
|
|
what is MOI?
|
multiplicity of infection: basically the amount of viable progeny viruses generated from one parent virus
|
|
|
occurring at a high MOI, the total yield of virions remains constant, but the number of infectious virions diminishes (due to defective mutations)
|
interference
|
|
|
viral genome becomes part of the cellular genome, and for example, activate Onc genes, causing tumor and cancer development in animals
|
integration
|
|
|
phenotypic mixing
|
during a mixed infection, the viral genome from one virus is enclosed in the capsid or envelope of another virus
|
|
|
interference
|
occurring at a high MOI, the total yield of virions remains constant, but the number of infectious virions diminishes (due to defective mutations)
|
|
|
integration
|
viral genome becomes part of the cellular genome, and for example, activate Onc genes, causing tumor and cancer development in animals
|
|
|
either the virus changed, allowing it to replicate without killing the cell or the cell changed, allowing it to replicate the virus without being lysed
|
persistence
|
|
|
intramolecular genetic exchanges between viruses, resulting in a new virus or variant
|
recombination
|
|
|
in viruses with segmented genomes, exchange of these gene segments to produce a new hybrid virus
|
reassortment
|
|
|
persistence
|
either the virus changed, allowing it to replicate without killing the cell or the cell changed, allowing it to replicates the virus without being lysed
|
|
|
recombination
|
intramolecular genetic exchanges between viruses, resulting in a new virus or variant
|
|
|
reassortment
|
in viruses with segmented genomes, exchange of these gene segments to produce a new hybrid virus
|
|
|
morphological changes of virally infected cells, such as rounding, lysis, detachment, syncytia, and inclusion bodies
|
cytopathic effect
|
|
|
cytopathic effect
|
morphological changes of virally infected cells, such as rounding, lysis, detachment, syncytia, and inclusion bodies
|
|
|
what are two causes of a cytopathic effect?
|
1. direct pathological injury of infected cells
2. side-effect: altered metabolism due to virus replication |
|
|
what comprises the VAP (viral attachment protein) of a:
- non-enveloped virus - enveloped virus |
- non-enveloped virus: part of the capsid or a protein extending from the capsid
- enveloped viruses: spike or peplomer glycoproteins in the envelope |
|
|
what host enzyme does a DNA virus use to transcribe its genome to mRNA in the nucleus?
|
DNA-dependent RNA polymerase II
|
|
|
which three families of RNA viruses replicate in the nucleus, as opposed to the cytoplasm?
|
1. Orthomyxoviridae
2. Coronaviridae 3. Retroviridae |
|
|
which types of viruses have infectious naked genomes?
|
- DNA viruses (except Poxviridae)
- (+) sense RNA viruses (except Retroviridae) |
|
|
what are four ways viruses inhibit cell metabolism in order to reproduce?
|
1. inhibit cellular transcription mechanisms (so cell will make viral mRNA)
2. inhibition of RNA processing pathways (so cell uses maturation pathways on viral mRNA) 3. inhibition of cellular translation (so viral proteins are encoded) 4. inhibition of host cell DNA synthesis (competition for NA precursors & redirect host cell enzymes for viral DNA synthesis) |
|
|
what are four ways that viruses change cell structure?
|
1. membrane fusion → syncytia
2. change permeability of the plasma membrane → cell lysis 3. disruption of cytoskeleton → rounding of the cells 4. incorporation of cytoskeletal components into infected cell structures → inclusion bodies |
|
|
multiple, continuous transmissions; disease presence in a defined population/region/time
|
enzootic
|
|
|
peaks in incidence exceeding the endemic (enzootic) baseline
|
epizootic
|
|
|
worldwide viral enzootics
|
panzootic
|
|
|
enzootic
|
multiple, continuous transmissions; disease presence in a defined population/region/time
|
|
|
epizootic
|
peaks in incidence exceeding the endemic (enzootic) baseline
|
|
|
panzootic
|
worldwide viral epizootics
|
|
|
number of new cases over number of subjects over a period of time
|
incidence or attack rate
|
|
|
continuous reporting of reportable diseases
|
surveillance
|
|
|
moment of infection to onset of clinical signs
|
incubation period
|
|
|
moment of infection to first day of virus shedding
|
generation time
|
|
|
from the first day to last day of virus shedding
|
period of infectivity
|
|
|
surveillance
|
continuous reporting of reportable diseases
|
|
|
incubation period
|
moment of infection to onset of clinical signs
|
|
|
generation time
|
moment of infection to first day of virus shedding
|
|
|
period of infectivity
|
from the first day to last day of virus shedding
|
|
|
what are six modes of transmission by viruses?
|
1. horizontal
2. vertical 3. zoonotic 4. vector-borne 5. iatrogenic 6. nosocomial |
|
|
which type of viruses usually use direct contact in horizontal transmission? Indirect contact?
|
- direct: enveloped
- indirect: non-enveloped |
|
|
virus integrated into the genome of the ovum, with transcription and replication in the offspring, which passes from generation-to-generation
|
Germline transmission
|
|
|
Germline transmission
|
virus integrated into the genome of the ovum, with transcription and replication in the offspring, which passes from generation-to-generation
|
|
|
what are four methods of viral vertical transmission?
|
1. transplacentally
2. perinatally 3. colostrum/milk 4. germline transmission |
|
|
what are five families of arboviruses?
|
1. Togaviridae
2. Flaviviridae 3. Bunyaviridae 4. Reoviridae 5. Rhabdoviridae |
|
|
what is the difference between a biological vector and a mechanical vector?
|
viruses in biological vectors replicate and magnify in the vectors, and have efficient transmission; viruses in mechanical vectors do not replicate, and are therefore not efficient for transmission.
|
|
|
viral diseases transmissible under NATURAL CONDITIONS from vertebrate animals to humans
|
zoonotic transmission
|
|
|
zoonotic transmission
|
viral diseases transmissible under NATURAL CONDITIONS from vertebrate animals to humans
|
|
|
true or false: most viral infections cause mild or asymptomatic diseases
|
true
|
|
|
what are five common routes of entry of viruses into the body?
|
1. respiratory tract
2. GI tract 3. conjunctiva 4. genitourinary tract 5. skin (epidermis) |
|
|
what are three types of viruses that commonly infect the host through a compromise in the skin?
|
1. Herpesvirus
2. Papillomavirus 3. Poxvirus |
|
|
why are non-enveloped viruses most suitable for GI tract infection? What is an example of an enveloped virus that infects the GI tract?
|
- the viral envelope is susceptible to extreme pH and enzymatic conditions to of the GI tract
- Coronaviridae are enveloped viruses that have evolved to favor the GI tract |
|
|
what is the difference between primary viremia and secondary viremia?
|
- primary: virus uses blood to transport from infection site to target replication sites
- secondary viremia: blood takes viruses replicated in organs to other sites, where it causes clinical manifestations |
|
|
what is the primary site of replication for viruses that spread via the lymphatic system?
|
epithelial cells, vascular endothelium
|
|
|
why does the immune system recognize that a cell is infected with a virus before progeny viruses are produced?
|
because of viral protein presentation by the MHC-I antigen
|
|
|
what are four ways in which the immune system destroys virus-infected cells and the basic mechanism?
|
1. Cytotoxic T cells: perforin
2. Ig-complex mediated cytotoxicity: low level of Ig via the Classical pathway 3. Ig dependent cell-mediated cytotoxicity: mediated by Fc receptors on phagocytes 4. NK cells: activated by IFN or viral glycoproteins, they non-specifically kill virus-infected cells |
|
|
what is the epitope for neutralizing antibodies?
|
viral attachment proteins
|
|
|
what are the two basic ways in which neutralizing antibodies kill viruses?
|
1. at attachment/entry stage, but this requires a large amount of antibodies
2. antibody-virus complexes destroyed by cellular enzymes: more effective |
|
|
which antibody provides mucosal immunity?
|
IgA
|
|
|
what are the most effective types of viral vaccines and why?
|
MLV. Because the vaccines are still infective, but not pathogenic. This allows for better immune development
|
|
|
what are the three basic roles of adjuvants?
|
1. prolonged retention and slow release of vaccine
2. macrophage activation 3. chemotactic for lymphocytes, leading to lymphokine secretion |
|
|
what is a chimeric virus?
|
a recombinant virus used in vaccines that has the antigens of one virus but the nucleic acid of a less pathogenic virus
|
|
|
comment on modified-live versus killed vaccines:
- route of administration - dose and cost - number of doses - use of adjuvant - duration of immunity - antibody class - development of CMI - heat stability - risk of reversion |
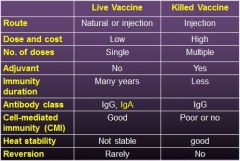
|

