![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
52 Cards in this Set
- Front
- Back
|
Ziegler-Natta catalyst
|
One of most important transition-metal catalysts in commerce, brings about polymerization of ethylene & other alkenes at 25 C & 1 atm
|
|
|
Reason Ziegler-Natta polymerization is better than free radical polymerization of ethylene
|
resulting high-density polyethylene has diff prop from low-density polyethylene prod
|
|
|
Mech Ziegler-Natta polymerization
|
Continuation of insertion-ligand association sequence gives polymer
|
|
|
It is believed that titanium brings about rxn because
|
D1 metal cant undergo B-elim (requires filled metal d orbitals) & tendency for B-elim of other metals would terminate rxn
|
|
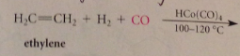
Hydroformylation
|
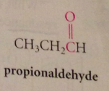
Tetracarbonylhydridocobalt(1) catalyst produces products like propionaldehyde: involves 1,2 insertion rxn of ethylene & 1,1 insertion rxn of CO
|
|
|
Homogeneous catalytic hydrogenation of alkenes
|
Uses soluble rhodium (I) catalyst called Wilkinson catalyst ClRh(PPh3)3
|
|
|
Phenoxide ion/ phenolate
|
conj base of phenol
|
|
|
Phenols are ___ acidic than alcohols
|

more: due to stabilization of conjugate-base anion due to resonance & polar effect of benzene ring (stabilized neg charge)
|
|
|
Substituent groups can affect phenol acidity by
|
polar & resonance effects: nitro group stabilizes conjugate base anion
|
|
|
P-nitrophenol is __ acidic than m-nitrophenol
|

more despite farther nitro group from phenol Oxygen: resonance (polar effects dec w distance)
|
|
|
M-nitrophenol has no resonance structure that delocalizes the neg charge into the nitro group
|
|
|
|
Acid strengthening resonance effect of O/P nitro groups so large that 2,4,6-trinitrophenol is
|
a strong acid
|
|
|
Factors that govern acidity
|
Element (bound to higher atomic #) in row, electroneg (h2o>methane, phenol> toluene) in column, rel bond E (thiols > ROH) charge (pos enhances) resonance, polar (stabiliz charge in conj b enhances)
|
|
|
Alcohols are not converted completely into alkoxides by aq. NaOH bc
|
pKa values water & alcohols are similar
|
|
|
Equilibrium for reaction of phenol & NaOH lies completely to the
|
right
|
|
|
Phenol treated with one equiv NaOH or NaOC2H5
|
phenol OH proton titrated completely to give solution of sodium phenoxide
|
|
|
Separate water-insoluble phenol 4-chlorophenol from water-insoluble alcohol 4-chlorocyclohexanol
|
Sodium or potassium salt like other alkali metal salts has considerable solubility in water bc ionic cmpd
|
|
|
When mixture phenol & alcohol in ether solution is shaken w aqueous NaOH,
|
phenol selectively extracted into aq solution as sodium salt while alcohol remains in ether
|
|
|
Acidification of aq solution gives
|
Phenol separate from solution bc after acidification, no longer ionized
|
|
|
Phenols are soluble in NaOH solution means that
|
if sodium hydroxide added
|
|
|
Qualitative test for phenols
|
Solubility in 5% NaOH & other cmpds of equal/greater acidity
|
|
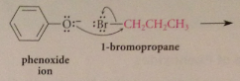
Can phenoxides be used as nucleophiles?
|
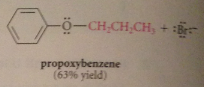
Yes
|
|
|
Quinones
|
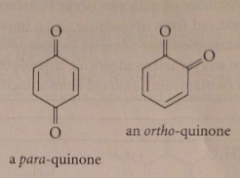
Most common oxidation products of phenols
|
|
|
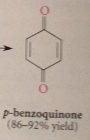
|
|
|
Names of quinones derived from
|
names of corresponding aromatic hydrocarbons (benzoquinone, napthoquinone)
|
|
|
Less stable quinone isomer
|
ortho-quinones: ends of C=O bond dipoles w like charges close together
|
|
|
Coenzyme Q
|
Oxidized form ubiquinone: in respiratory chain localized in mitochondrion, converts O2 into H2O & harnesses E released to synth ATP
|
|
|
Doxorubicin
|
isolated from a microorganism: important antitumor drug
|
|
|
Oxidation of phenols by air
|
causes darkening when some phenols stored for time
|
|
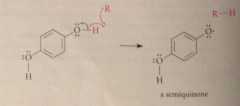
Practical applications of phenol oxidation
|
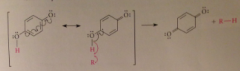
Inhibit free-radical rxns resulting in oxidation of other cmpds (semiquinone resonance stabilized) then hydroquinone terminates free rad chain rxns by intercepting free-rad intermed & reducing to RH + effectiveness of several widely used food preservatives
|
|
|
How does food discolor and spoil and how does preservative like BHT work?
|
Oxidation involving free-radical processes -- BHT donates OH hydrogen atom to freee radicals => phenoxy radical too unstable to & unreactive to propagate radical chain reactions
|
|
|
Vitamin E
|
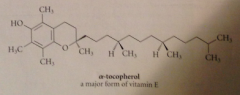
Phenol, major compd in blood prevents oxidation damage by free radicals: acts by terminating radical chains
|
|
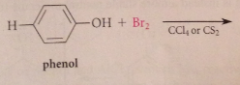
EAS of phenols
|
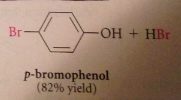
OH sometimes has special effects not common (bc strongly activating substituent, phenol can be halogenated once under mild conditions)
|
|
|
|
|
|
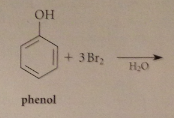
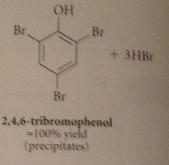
Extensive bromination occurs bc
|
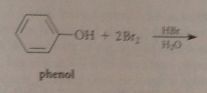
bromide reacts w h2o to give protonated hypobromous acid, more electrophile than bromine, then partially ionizes to conj base phenoxide anion: very reactive (not carbocation but neutral mlc), brominates instantly, pulls phenol-phenolate equil right
|
|
|
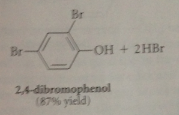
P-bromophenol is in equilibrium with conjugate base p-bromophenoxide anion, which brominates again until all o/p positions substituted
|
Second & third sub, powerful o/p directing & activating effects of O- group override weaker of bromine substituents
|
|
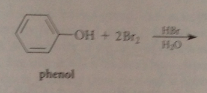
|
|
|
|
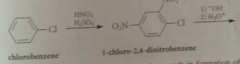
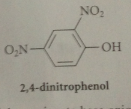
Phenol is very reactive in EAS such as
|
nitration (once under mild conditions)
|
|
|
|
|
|
The basic conditions of the reaction result in formation of the conjugate-base anion of the prod
|
H3O added for neutral phenol
|
|
|
The great reactivity of phenol is EAS doesn't extend to FC acylation bc
|
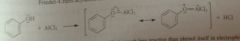
phenol reacts rapidly with AlCl3 catalyst: adduct is much less reactive than phenol itself in EAS bc O2 delocalized onto Alum. = less available for resonance stabilization of carbocation
|
|
|
FC acylation/alkylation of phenol
|
occurs slowly but can be carried out successfuly at high temp. not highly activated so ring only acylated once
|
|
|
Carbon-oxygen reactivity of phenols
|
Follows poor carbon-halogen reactivity of aryl halides: do not undergo SN1 or E1 reactions for same reasons & phenols do not react under conditions used for Sn1/E1 of alcohols
|
|
|
Phenols plus conc HBR
|
no rxn
|
|
|
Phenols plus conc H2So4
|
no dehydration, but sulfonation
|
|
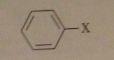
where X is a good LG
|
no reactivity toward SN1/Sn2 conditions
|
|
|
In aryl ethers, cleavage occurs only
|
at alkyl-o2 bond (1 set products)
|
|
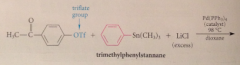
Stille Reaction: Aryl triflates with organotin derivatives in presence of Pd(0) catalyst
|
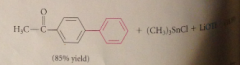
Coupling products
|
|
|
transferred preferentially
|
Vinylic, aryl groups, other unsat groups
|
|
|
If tetraalkylstannane is used
|
alkyl groups can be transffered (not plagued by rearrangement)
|
|
|
Mechanism Stille reaction
|
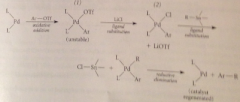
Oxidative addition of aryl triflate to 14e Pd(PPH3)2 = unstable complex, excess chloride ion rescues from decomposition by ligand substitution, R group on organotin compds have carbanion character, nucleophilic substitute for chloride on Pd => reductive elimination
|
|
|
Principle method used to prepare phenol
|
cumene from petroleum produces phenol and acetone through autoxidation (O2 is oxidation agent) and then acid-catalyzed rearrangement
|

