![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
106 Cards in this Set
- Front
- Back
|
H at a-C of carbonyl cmpds are
|
acidic
|
|
|
When an a-proton is removed
|
a conj B anion is formed @ a-C
|
|
|
a-C of a carbonyl cmpd, as site of conj B enolate ion is site of
|
nuc reactivity
|
|
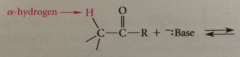
conj B anion of a carbonyl cmpd formed by removal of an a-H
|
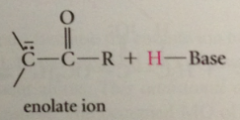
enolate ion
|
|
|
enol
|
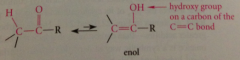
any cmpd in which hydroxy group is on a C of a C-C db (vinylic alcohol) -- corresponding carbonyl cmpd is constitutional isomer
|
|
|
most carbonyl cmpds w a-H are in equil w small
|
amts of enol isomers
|
|
|
despite low conc, enols are
|
intermediates in important rxns of carbonyl cmpds
|
|
|
a, b -unsat carbonyl cmpds
|
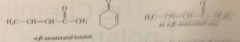
carbonyl group conj w C-C db
|
|
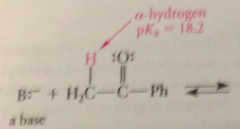
ionization of an a-H gives conj B
|
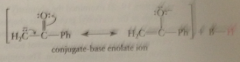
enolate anion
|
|
|
pKa values of simple aldehydes/ketones are in the range
|
16-20
|
|
|
pKa values of esters
|
prob within few units of 25
|
|
|
a-H of nitriles/tertiary amides
|
acidities similar to those of esters
|
|
|
although carbonyl cmpds are weak A
|
a-H more acidic than other types of H bound to C
|
|
|
Stabilization of a base __ the pKa of its conj A
|
lowers
|
|
|
Why do carbonyl cmpds have a lower pKa value
|
resonance stabilized
|
|
|
resonance
|
symbolic way of depicting orbital overlap
|
|
|
hybridization of enolate ion
|
anion a-C is sp2 hybridized: allows unshared pair of e to occupy a 2p orbital, aligned for overlap w 2p orbitals of carbonyl group
|
|
|
2p orbital alignment for conj B enolate ion acetaldehyde
|
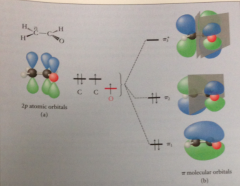
results in formation of 3 pi MOs, 2 filled by pi e
|
|
|
Occupied MO of lowest E
|
pi e extend across all 3 constituent atoms: additional overlap provides additional bonding ~ stabilization
|
|
|
Occupied MO of higher energy pi2 has node @ carbonyl C
|
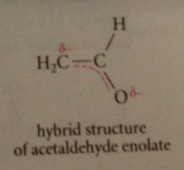
e involved in chem rxns of eno late ions - a-C & carbonyl O are major sites of e density
|
|
|
if the structure of an enolate ion constrains geometry of component 2p orbital so cannot overlap
|
enolate ion no longer stabilized
|
|
|
delocalization reason for acidity of a-H
|
neg charge delocalized onto O (electroneg) so a-H much more acidic than allylic H of alkenes altho conj B anions of both types resonance-stabilized
|
|
|
polar effect of carbonyl group
|

stabilized enolate anions like carboxylate anions - results from favorable interaction of pos end of C=O bond dipole w neg charge of ion
|
|
|
difference in acidity btwn aldehydes, ketones & esters
|
delta G = 2.3RT(pKa) if G of carbonyl lowered relative to conj B enolate, G inc, pKa inc, acidity reduced. G of ester loewred relative to ketone by resonance interaction of ester O w carbonyl group - resonance effect overrides polar effect of ester O, which in absence of resonance would inc acidity of esters relative to ketones. in enolate ion, analogous resonance structure much less important bc of repulsion btwn neg charges, so loss of most ester resonance on ionization inc G for ionization of ester
|
|
|
Are amide N-H hydrogens a-hydrogens?
|
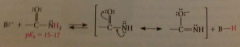
Yes, they are attached to an atom that is adjacent to a carbonyl group
|
|
|
are carboxylic OH hydrogens a-H?
|
Yes -
|
|
|
Acidity order
|
CA > amides > aldehydes, ketones because of electroneg element effect
|
|
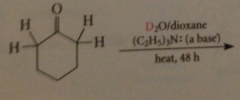
enolate ions are bronsted B - only H that can be exchanged for deuterium by treating the carbonyl cmpd w base in D2O
|
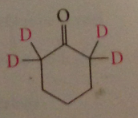
a-hydrogens of an aldehyde or ketone and no others
|
|
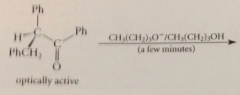
if an optically active aldehyde or ketone owes chirality solely to asymm a-C & if C bears a H
|
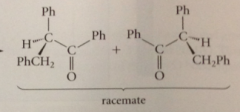
cmpd will be racemized by base
|
|
|
why does racemization occur?
|
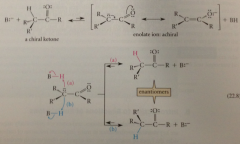
enolate ion is achiral bc of sp2 hybridization @ anionic C (ionic a-C & attached groups lie in one plane, anion can be reprotonated at either face) occurs relatively quickly if carbonyl cmpd is left in contact w B
|
|
|
a-H exchange & racemization rxns of aldehydes & ketones occur
|
more readily than those of esters bc aldehydes & ketones are more acidic than estres, so form enolate ions more rapidly, under milder conditions
|
|
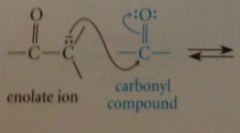
|

enolate ions react as nuc @ carbon of carbonyl groups
|
|
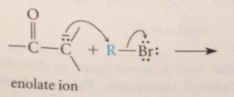
enolate ions as lewis B react w alkyl halides & sulfonate esters
|
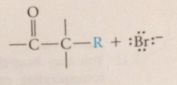
|
|
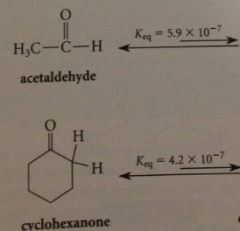
carbonyl cmpds w a-H are in equil w small amt enol isomers
|
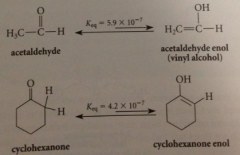
unsymm ketones are in equil w 1+ enol
|
|
|
esters contain more or less enol isomers than aldehydes or ketones?
|
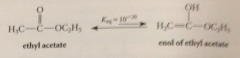
less
|
|
|
most carbonyl cmpds are
|
more stable than corresponding enols
|
|
|
interconversion of enols & corresponding carbonyl cmpds catalyzed by
|
A & B - slow in dilute soln in absence of A/B catalysts
|
|
|
enolizations of esters & CA are __ favorable than enolizations of aldehydes & ketones
|
less
|
|
|
The major reason for the instability of enols is
|
C=O db of carbonyl is stronger than C=C db of enol
|
|
|
in esters and acids, additional instability of enols results from
|
loss of stabilizing resonance interaction btwn carboxylate O & carbonyl pi e in carbonyl forms
|
|
|
are some enols more stable than corresponding carbonyl cmpds?
|
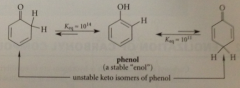
Yes - phenol is conceptually an enol, a vinylic alcohol, but is more stable than its keto isomers bc phenol is aromatic
|
|
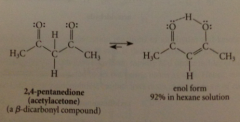
Enols of B-dicarbonyl relatively stable bc 2 carbonyl groups separated by 1 C
|
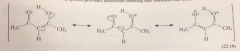
conjugated, resonance stabilization (pi e overlap) provides add bonding that stabilized enol, intramlclr H bond
|
|
|
formation of enols & of enols into carbonyl cmpds are catalyzed by
|
A & B
|
|
|
enols are diff. to isolate as pure cmpds under most ordinary circumstances bc
|
rapid conversion into carbonyl cmpds
|
|
|
enolization
|
conversion of carbonyl cmpd into enol
|
|
|
base-catalyze enolization
|
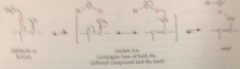
intermediate enolate ion (consequence of acidity of a-H) protonation of enolate anion by water on a-C gives back carbonyl cmpd; protonation on O gives enol
|
|
|
acid-cat enolization
|
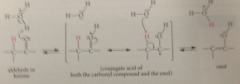
conj A of carbonyl cmpd (carbocation) - loss of proton from O gives back starting carbonyl cmpd, loss from a-C gives enol
|
|
|
enolate ion is
|
conj B of both carbonyl cmpd & enol
|
|
|
enol & carbonyl isomer have ___ conj A
|
the same
|
|
|
exchange of a-H for deuterium & racemization at a-C catalyzed by
|
bases & acids
|
|
|
acid-cat processes can be explained by
|
intermediacy of enols - formation of carbonyl cmpd from enol introduces H from solvent @ a-C, accounts for observed isotope exchange (C of an enol is not asymm, so racemization observed in acid)
|
|

halogenations of aldehyde/ketone in acidic soln usually results in
|

replacement of one a-H by halogen
|
|
|
reactive intermediates
|

enols
|
|
|
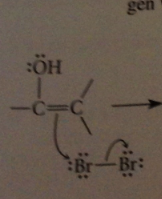
do enols react w halogens?
|

yes, but only add one halogen atom to db, then resulting carbocation intermediate loses proton instead of adding to second halogen
|
|
|
carbocation destabilized by unfavorable interaction with
|
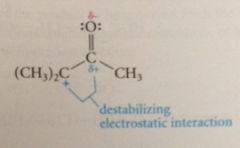
bond dipole of carbonyl group
|
|
|
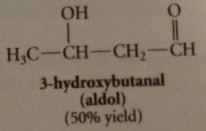
|
|
|
aldol
|
3-hydroxybutanal / b-hydroxy aldehydes
|
|
|
aldol addition
|
rxn of 2 aldehyde mlcs to form b-hydroxy aldehyde
|
|
|
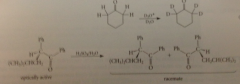
base-catalyzed aldol addition involves
|
enolate ion as intermediate
|
|
|
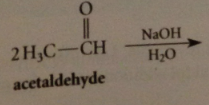
enolate ion formed by rxn of acetaldehyde w aqueous NaOH
|
adds to a 2nd mlc of acetaldehyde
|
|
|
is aldol addition a nuc addition to a carbonyl group?
|
yes
|
|
|
|
|
|
how is aldol addition related to cyanohydrin formation?
|

nucleophilic addition
|
|
|
is aldol addition reversible?
|
yes
|
|
|
equil for aldol addition is
|
more favorable for aldehydes than ketones
|
|
|
the isotope exchange is accounted for by
|
enol introduces H from solvent @ a-C
|
|
|
C of an enol, like an enolate ion is
|
not asymmetric, accounts for racemization in acid
|
|
|
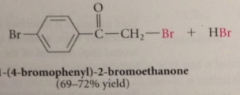
|
|
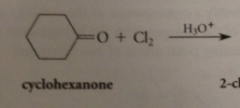
|
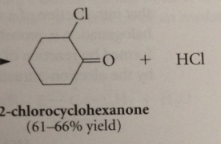
|
|
|
reactive intermediates
|
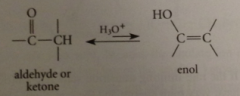
enols
|
|
|
acid-cat halogenation rate law
|
rate = k[ketone][H3O+] -- independent of halogen conc
|
|
|
RL process in acid-cat halogenation of aldehydes & ketones
|
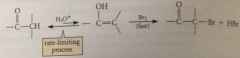
enol formation (RLS is 2nd: removal of a-proton)
|
|
|
why is intro of 2nd halogen much slower than first?
|
bc only one halogen intro at given a-C in acidic soln
|
|
|
slower halogenation probably consequence of
|
stability of carbocation intermediate formed by rxn of halogen w halogenated enol
|
|
|
carbocation destabilized by e-attracting polar effect of
|
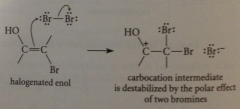
2 halogens (TS high energy, small rate)
|
|
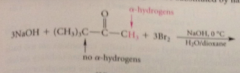
halogenation of aldehydes & ketones w a-H also occurs in base
|
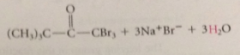
all a-H substituted by halogen
|
|
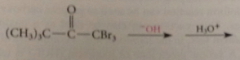
when aldehyde or ketone sm either acetaldehyde or methyl ketone
|
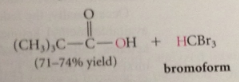
prod of halogenation is trihalo carbonyl cmpd, not stable under rxn conditions, reacts further to give after acidification of rxn mixture CA and a haloform
|
|
|
haloform rxn
|
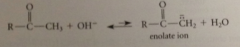
conversion of acetaldehyde / methyl ketone into CA & haloform by halogen in base followed by acidification
|
|
|
enolate ion reacts as
|

nuc w halogen to give a-halo carbonyl cmpd
|
|
|
second bromination occurs bc
|
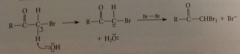
enolate ion of a-halo ketone formed more rapidly than enolate ion of starting ketone - polar effect of halogen stabilizes enolate ion & by Hammond's postulate, TS for enolate-ion formation
|
|
|
dihalo carbonyl cmpd
|
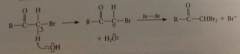
brominates even more rapidly
|
|
|
C-C bond broken when
|
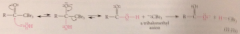
trihalo carbonyl cmpd undergoes nuc acyl sub rxn
|
|
|
LG is
|
trihalomethyl anion - carbanions too basic to serve as LG but trihalomethyl anions less basic
|
|
|
basicity of trihalomethyl anions low enough for LG but high enough to
|
react irreversibly w CA byproduct
|
|
|
CA can be isolated by
|
acidifying the rxn mixture
|
|
|
iodoform test
|
haloform rxn used to prepare CA from readily available methyl ketones - qualitative test for methyl ketones
|
|
|
how does iodoform test work
|
cmpd of unknown structure mixed w alkaline I2, yellow ppt of iodoform (HCI3) taken as evidence for methyl ketone
|
|
|
why is iodoform test specific for methyl ketones?
|
only by replacement of 3 H w halogen does C become good enough LG for nuc acyl sub rxn
|
|
|
alcohols that give positive iodoform test
|
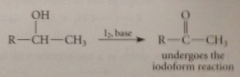
oxidized to methyl ketones (or acetaldehyde for ethanol) by basic iodine soln
|
|
|
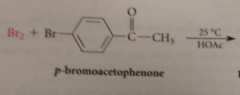
CA can br brominated @
|
a-C
|
|
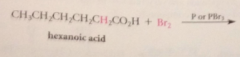
Hell-Volhard Zelinsky rxn aka HVZ
|
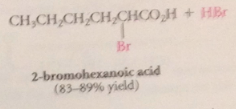
|
|
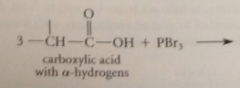
first stage in mech of HVZ
|
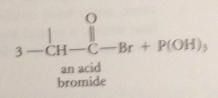
conversion of small amt CA into acid bromide by catalyst PBr3
|
|
|
mechanism closely resembles that for acid-cat bromination of ketones
|
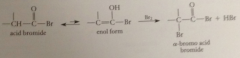
enol is species that actually brominates
|
|
|
when small amt PBr3 cat used
|

a-bromo acid bromide reacts w CA to form more acid bromide, which is then brominated (a-bromo acid formed)
|
|
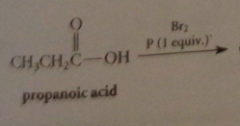
if one full equiv PBr3 used
|

a-bromo acid bromde is rxn prod - can be used in many rxns of acid halides
|
|
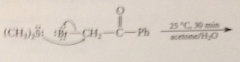
most a-halo cmpds are very reactive i sn2
|
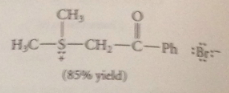
used to prepare other a-sub carbonyl cmpds
|
|
|
for a-halo ketones, nuc must not be too
|
basic
|
|
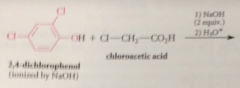
|
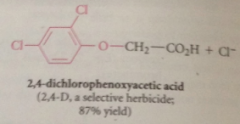
|
|

|

|
|
|
enolate-ion formation promoted by
|
stronger bases
|
|
|
more basic nuc can be used w
|
a-halo acids bc under basic conditions a-halo acids are ionized to form carboxylate conj B anions - 2nd ionization to give enolate ion would intro 2nd neg charge, doesn't occur
|
|
|
a-halo carbonyl cmpds more reactive bc
|
same reason inc reactivity allylic alkyl halides in sn2 displacements
|
|
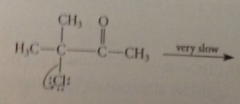
a-halo carbonyl cmpds react so slowly by sn1
|
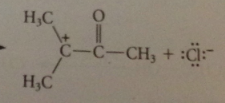
this rxn not useful
|
|
|
rxns that require formation of carbocations alpha to carbonyl groups
|
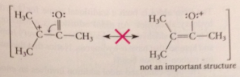
generally do not occur (an a-carbonyl cmpd should be resonance stabilized, but its resonance structure is not important)
|
|
|
carbocation also destabilized by
|
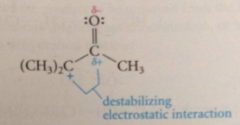
unfavorable electrostatic interaction w bond dipole of carbonyl group (partial pos charge on carbonyl C atom)
|
|
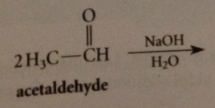
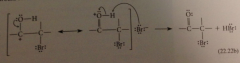
aldol addition
|
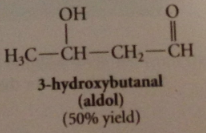
|

