![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
59 Cards in this Set
- Front
- Back
|
General Addition Reaction of Alkenes:
|
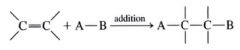
|
|
|
What are the two characteristics of the double bond that helps us understand how addition reactions occur?
|
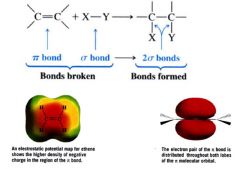
1. An addition reaction results in the conversion of one pi bond and one sigma bond into two sigma bonds. (usually energetically favorable b/c pi bonds are weaker... usually exothermic)
2. e-s of the pi bond are exposed. B/c the pi bond results from overlapping p orbitals, the pi e-s lie above and below the plane of the double bond. |
|
|
Electrophiles:
|
Electron-seeking reagents.
--- include: - (+) reagents such as H+ - neutral reagents such as bromine (b/c it can be polarized so one end is (+)) - lewis acids such as BH3, BF3, AlCl3, metal ions that contain vacant orbitals... etc. |
|
|
Nuclophile:
|
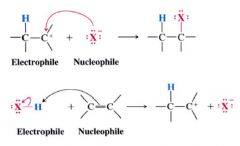
Molecules/ ions that can furnish an e- pair (ex: lewis bases)
|
|
|
General hydrogen halide addition to the double bond of a alkene:
|
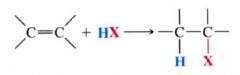
|
|
|
How the reaction of an addition of a hydrogen halide to an alkene occurs:
|
- by dissolving the H-X in a solvent such as acetic acid, or CH2Cl2
- by bubbling the gaseous H-X directly into the alkene and using the alkene itself as the solvent. |
|
|
The order of reactivity of hydrogen halides:
|
HI > HBr > HCl > HF
- unless alkene is highly subsituted, HCl reacts so slowly that it is hardly ever used. |
|
|
Markovnikov's Rule:
|
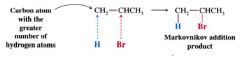
in the addition of HX to an alkene, the H atom adds to the C atom of the double bond that already has the greatest # of H atoms
|
|
|
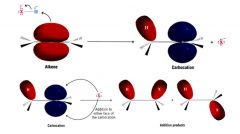
A mechanism for the reaction: Addition of a H-X to an alkene:
|

|
|
|
Energy diagram for the addition of a HX to an alkene reaction:
|
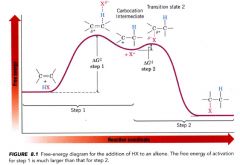
Step 1:
- rate determining step - alkene donates a pair of e-s to the proton of HX & forms a carbocation - highly endergonic - has high free energy of activation - takes place slowly step 2: - highly reactive carbocation stabilizes itself by combining with halide ion - exergonic - low free energy of activation - takes place quickly. |
|
|
A mechanism for the reaction: Addition of HBr to 2-Methylpropene
|
|
|
|
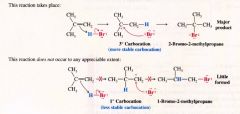
The Modern statement of Markovnikov's Rule:
|
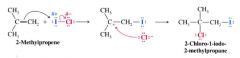
In the ionic additon of an unsymmetrical reagent to a double bond, the positive portion of the adding reagent attaches itself to a carbon atom of the double bond so as to yield the more stale carbocation as an intermediate [determines the overall orientation of the reaction]
|
|
|
Regioselective:
|
When a reaction that can potentially yield 2/ more constitutional isomers actually produces only one (or a predominance of one)
|
|
|
Anti-Markovnikov addition:
|
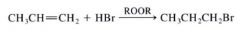
When the H atom becomes attached to the C atom w/ the fewer H atoms.
ex: The addition of HBr to alkenes in the presence of periodixes (ROOR) |
|
|
The Stereochemistry of the reaction: Ionic Addition to an Alkene:
|
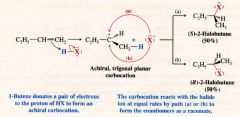
|
|
|
The addition of Sulfuric acid to an alkene:
|
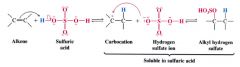
Alkenes dissolve b/c they react by electrophilic additon to form alkyl hydrogen sulfates.
- is regioselective - follows Markovnikov's Rule |
|
|
Creating alcohols from Alkyl Hydrogen Sulfates:
|
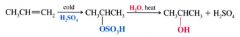
Alkyl hydrogen sulfates can be easily hydrolyzed to alochols by heating them w/ water.
- Markovnikov addition of H- and -OH |
|
|
A Mechanism for the reaction: Acid-Catalyzed Hydration of an Alkene:
|
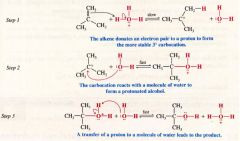
- reaction follows markovnikov's rule
|
|
|
Oxymercuration- Demercuration:
|
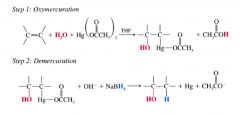
- synthesizing alcohols from alkenes that avoids rearrangements
- is hignly regioselective, follows Markovnikov's rule 2-step method: step 1: OXYMERCURATION: water and mercuric acetate add to the double bond step 2: DEMERCURATION: sodium borohydride reduces the acetoxymercury group & replaces it w/ H. |
|
|
Does rearrangements occur in oxymercuration-demercuration?
|
is not prone to hydride or alkanide rearrangements.
|
|
|
A Mechanism for the reaction: Oxymercuration:
|

|
|
|
Alchols from alkenes through hydroboration-oxidation: Anti-Markovnikov Syn Hydration:
|
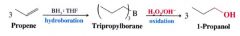
|
|
|
A Mechanism for the Reaction: Hydroboration:
|
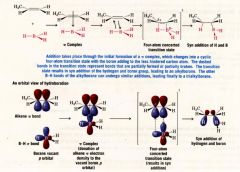
- in each addition step the boron atom becomes attached to the less sub. C atom of the double bond, and H atom is transferred from the boron atom to the other C atom of the double bond
- due to electronic and steric factors (the bulky boron-containing group can approach the less sub. C atom more easily) |
|
|
The stereochemistry of Hydroboration:
|
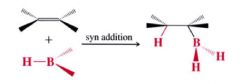
|
|
|
A Mechanism for the Reaction: Oxidation of Trialkylboranes (oxidation and hydrolysis of alkylboranes)
|
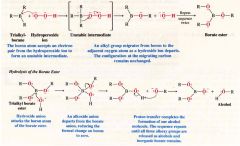
- regioselective
- net reslt is anti-Markovnikov addition of water - gives us a method for preparation of alcohols that cannot normally be obtained through the acid-catalyzed hydration of alkenes or by oxymercuration-demercuration. - the hydroxyl group replaces the B atom where it stands in the alkylboron compound. - net results is the syn addition of -H and -OH. |
|
|
Protonolysis of Alkylboranes:
|
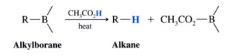
heating an alkylborane w/ acetic acid causes cleavage of the C-B bond and replacement w/ H
- H replaces B where it stands in the alkylborane. The stereochemistry of this reaction therefore is SYN. |
|
|
Bromine test detecting alkenes, and alkynes:
|
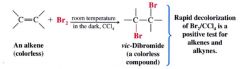
If bromine is added to an alkene (or alkyne) the red-brown color of the bromine disappears almost instantly as long as the alkene (or alkyne) is present in excess.
|
|
|
Mechanism for the Reaction: Addition of Bromine to an Alkene:
|

- an SN2 reaction
|
|
|
What occurs when a "cyclo" is attacked with bromine?
|
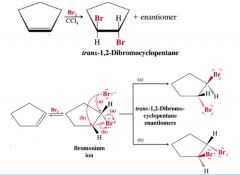
anti-addition occurs and product is a racemic mixture. A bromide ion attacks a C atom of hte ring from the side opposte that of the bromonium ion.
- SN2 reaction - The nucleophilic attack causes inversion of the congiguraiton of the carbon being attacked. |
|
|
How inversion occurs in the reaction of cyclohexan with bromine:
|
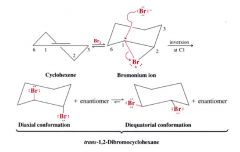
|
|
|
Stereospecific reaction:
|
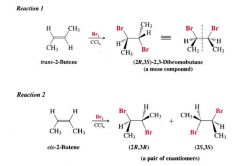
a reaction is stereospecific when a particular stereoisomeric form of the starting material reacts in such a way that it gives a specific stereoisomeric form of the product.
|
|
|
A Stereochemistry of the reaction: Addition of Bromine to cis- and trans-2-butene:
|
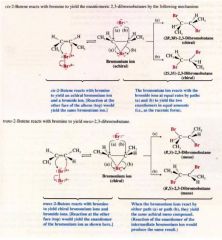
|
|
|
A Mechanism for the reaction: Halohydrin Formation from an Alkene:
|
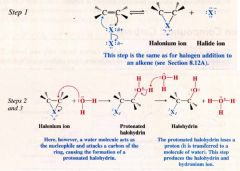
If the alkene is unsymmetrical, the halogen ends up on the carbon atom with greater # of H atoms.
|
|
|
Carbenes:
|
group of compounds in which carbon forms only two bonds.
- most are highly unstable - soon after they are formed, they usually react w/ another molecule. ---- reactions usually show a remarkable degree of stereospecificity. |
|
|
Methylene:
|
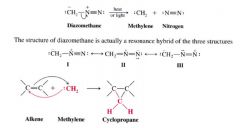
- the simplest carbene
- can be prepared by decomposition of diasomethane (CH2N2) a very posionous yellow gas. ----- can be accomplished by... 1. heating (thermolysis) 2. irradiating it with light of wavelengths that can absorb (photolysis) METHYLENE REACTS W/ ALKENES BY ADDING TO THE DOUBLE BOND TO FORM CYLOPROPANES |
|
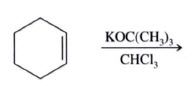
|
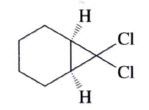
7,7-Dichlorobicyclo[4.1.0]heptane (59%)
|
|
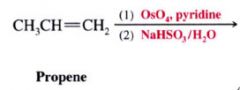
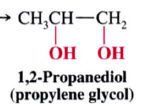
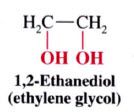
1,2-Dihydroxylation:
|
|
|

|
|
|
|
Mechansim for syn dihydroxylation of alkenes:
|
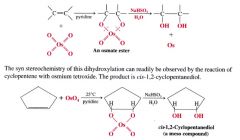
- involves a cyclic intermidate that results in SYN ADDITION of the O atom
- cleavage @ the oxygen-metal bonds take place w/o altering the stereochemistry of the 2 new C-O bonds. |
|
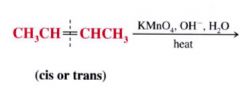
Cleavage w/ hot basic Potassium Permanganate
|
|
|
|
Using Potassium Permanganate (KMnO4) to test for alkenes/ alkynes:
|
it is a chemical test for the presence of unsaturations in an unknown compound. Solutions of potassium permanganate are purple... if an alkene / alkyne is present the purple color is discharge and a brown precipitate of manganese dioxide (MnO2) forms as the oxidation takes place.
|
|
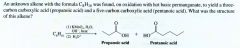
|

|
|
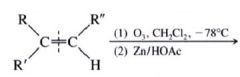
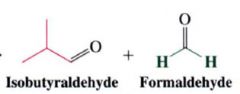
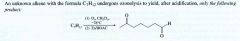
Ozonolysis:
|
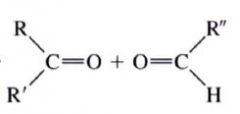
- the most useful method for cleaving alkenes.
- consits of bubbling ozone into a very cold (-71 degree C) soln. of the alkene in CH2Cl2, followed by treatment of the soln w/ zinc and acetic acid (or dimethyl sulfide) |
|

ozonolysis:
|
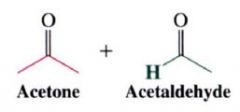
|
|
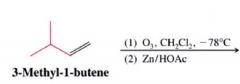
|
|
|
|
|
|
|
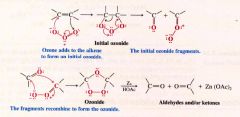
A Mechanism for the reaction: Ozonolysis of an Alkene
|
|
|
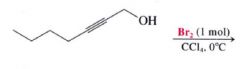
|
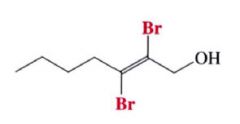
- the addition may occur once or twice, depending on the # of molar equivalents of halogen that are employed
- Most additions of Cl & Br to alkynes are anti additions and yield trans-dihaloalkenes. |
|
|
Addition of hydrogen halides to alkynes in general:
|
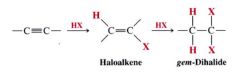
- regioselective
- folow markovnikov's rule |
|

|
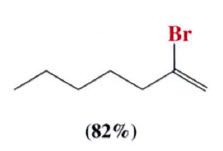
- follows Markovnikov's rlue
|
|

|
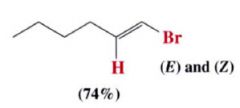
Anti-Markovnikov addition
|
|
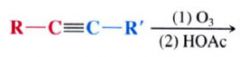
|
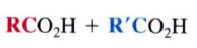
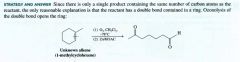
cleavage at the Carbon carbon triple bond.
|
|
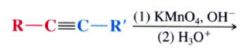
|
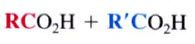
cleavage at the carbon-carbon triple bond.
|
|
|
When planning a synthesis what must be considered (4 interrelated aspects):
|
1. construction of the carbon skeleton
2. functional group interconversions 3. control of regiochemistry 4. control of stereochemistry. |
|
|
Stereoselective Reaction:
|
one in which the reactant is not necessarily chrial but in which the reaction produces predominantly or exclusively one stereoisomeric form of the product (or a certain subset of stereoisomers from among all those that are possible)
|
|
|
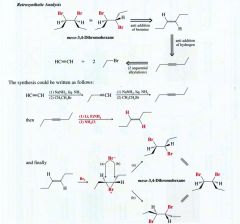
Stereospecific Reaction:
|
one that produces predominantly/ exclusively one stereoisomer of the product when a specific stereoisomeric form of the reactant is used.
|
|
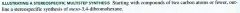
|
|
|
|
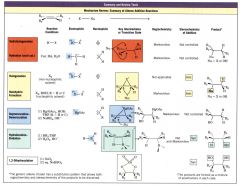
Summary and Review Tools: Mechansim Review: Summary of Alkene Addition Reactions
|
|
|
|
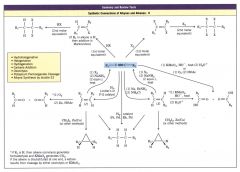
Summary and Review Tools: Sunthetic Connections of Alkynes and Alkenes: II
|
|

