![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
26 Cards in this Set
- Front
- Back
|
Hemoglobin |
– Globin protein bound to a hememolecule containing iron – Usually within blood cells – Appears red when oxygenated – Myoglobin is a type ofhemoglobin found in muscles |
|
|
Hemoglobin saturation in lungs and tissues- Partial pressures in mmhg needed for % saturation |
–Alveolar levels of 100 mmHg giveshemoglobin saturation • Alveolar levels of 60 mmHg gives 90%hemoglobin saturation (high altitudes,compromised pulmonary ventilation) –Tissue levels of 40 mmHg – hemoglobinsaturation is 75% • active tissue oxygen levels of 20 mmHg –hemoglobin saturation is 35% |
|
|
PO2 curve for O2 bound heme groups and % concentration in blood (line shape) |
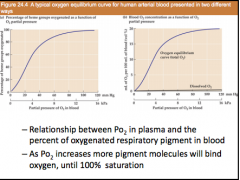
|
|
|
Hemoglobin affinity and relation to human development |
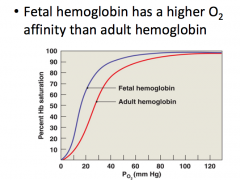
|
|
|
In what three ways does activity effect tissues? |
• Partial pressure of oxygen ‐ PO2 – Levels of PO2 decrease as tissue becomes more active • pH level – More acidity in tissues as they become moreactive • Temperature – Temperatures increase in active tissues |
|
|
Bohr Effect and root effect |
– Bohr effect or shift • Decrease in pH or increase in PCO2 reduces oxygenaffinity; “right shift” • P50 is increased • Facilitates oxygen transport to active tissues andfacilitates oxygen binding at the respiratory surfaces – Root effect• A Bohr effect with a reduction in the oxygen carryingcapacity |
|
|
Bohr Effect (graph) |
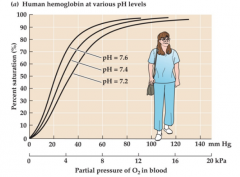
|
|
|
In what ways is the affinity of O2 affected |
• Temperature– Increases in temperature decrease oxygen affinity;“right shift”– P50 is increased • pH & CO2– Decrease in pH and increase in CO2 decreaseaffinity; “right shift” • Organic modulators (e.g., DPG)– Increases in these moddulators decrease oxygenaffinity; “right shift” |
|
|
Taxonomic variation of gas transport (diffusion vs blood) |
– Sponges, cnidarians, echinoderms, and insectscirculate external medium (water or air) pastalmost every body cell and can rely on diffusion– Larger animals use circulatory systems• Transport of gases in blood |
|
|
Respiratory pigments- function and need |
• Solubility of oxygen in aqueous fluids is low • Metalloproteins (respiratory pigments)– Proteins containing metal ions which reversiblybind to oxygen and – Increase oxygen carrying capacity by 50‐fold – Three major types of respiratory pigments • Hemoglobins– Chlorocruorins • Hemocyanins • Hemerythrins |
|
|
Hemocyanins (taxonomic groups, metal binder, where it is found in the body, color when oxygenated) |
• Arthropods and molluscs • Contain copper directly bound to protein (no heme) • Usually dissolved in the hemolymph • Appears blue when oxygenated |
|
|
Hemerythrins (taxonomic groups, metal binder, where it is found in the body, color when oxygenated) |
• Sipunculids, priapulids, brachiopods, some annelids • Contain iron directly bound to protein (no heme) • Usually found inside coelomic cells • Appears violet‐pink when oxygenated |
|
|
Shapes of O2 concentration curves for myoglobin and hemoglobin and why they are shaped that way |
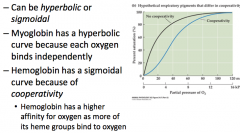
|
|
|
Root effect (graph) |
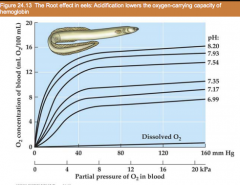
|
|
|
Relation between P50 O2 and diphosphoglycerate (DPG) |
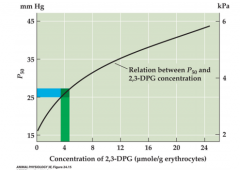
|
|
|
O2 transport in trout (graph) -relation of arterial and venous O2 concentration -warm vs cold acclimatized |
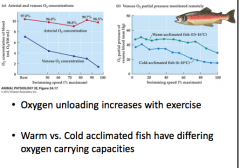
|
|
|
Blood O2 delivery in cephalopods |
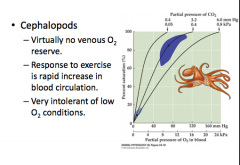
|
|
|
Allometric relationship and hemoglobin affinity |
• Allometry & hemoglobinaffinity – Smaller species havehemoglobin with loweroxygen affinity – Favors oxygen unloadingand higher weight‐specificmetabolic rates |
|
|
hypoxia acclimation in water fleas |
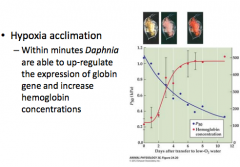
|
|
|
Three ways CO2 is transported in Blood |
– Small amounts of CO2 gas are transported in theplasma• CO2 is more soluble in body fluids than O2 – Some CO2 binds to proteins• For example, carbaminohemoglobin – Most CO2 is transported as bicarbonate (HCO3–) CO2 + H2O<-->H2CO3(carbonic acid)<-->HCO3- (bicarbonate) + H+ • Carbonic anhydrase catalyzes the formation of HCO3– |
|
|
Haldane effect |
• Deoxygenated bloodcan carry more CO2than oxygenated blood • The formation of carbaminohemoglobin isfavored on deoxygenated hemoglobin • Deoxygenated hemoglobin is a better protonacceptor than the oxygenated form -Allows for transport of H+ (proton in hemoglobin), and carbaminohemoglobin |
|
|
creation of HCO3- and release |
-created by carbonic anhydrase in the red blood cells -Is then exchanged into the plasma through Cl- ion (to balance charge) -At respiratory surface transforms back into carbondioxide and water HCO3- + H+ <--> CO2 +H2O |
|
|
diagram of CO2 transport |
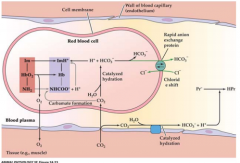
|
|
|
Haldane effect (graphs) |
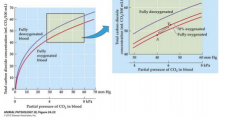
|
|
|
Relation between PCO2, pH and HCO3- |

|
|
|
blood pH and temperature |
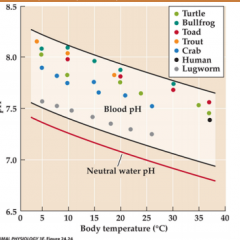
|

