![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
19 Cards in this Set
- Front
- Back
|
Name the three types of particle in an atom. State the relative mass and charge of each particle.
|
>Proton - mass 1, charge +1
>Neutron - mass 1, charge 0 >Electron - mass 1/1840, -1 |
|
|
How are the particles in an atom arranged?
|
Protons and neutrons in central nucleus held by strong nuclear force. Electrons orbit nucleus in shells (electrostatic force).
|
|
|
How many electrons do the first shells hold?
|
2n squared / n = number of shell
1st = 2 2nd = 8 3rd = 18 |
|
|
Write the shorthand for 9 electrons.
|
2,7,
|
|
|
Draw the electron arrangement diagram of an atom with 16 electrons.
|
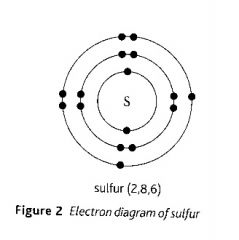
|
|
|
Define the term mass number, A.
|
The sum of the protons and neutrons in the nucleus of an atom.
|
|
|
Define the term atomic number, Z.
|
The number of protons in the nucleus of an atom.
|
|
|
Define the term Isotope.
|
Atoms with the same number of protons but different numbers of neutrons.
|
|
|
Why do isotopes of the same element have identical chemical properties?
|
They have the same number and arrangement of electrons.
|
|
|
What does a mass spectrometer measure?
|
Relative atomic masses
Relative molecular masses |
|
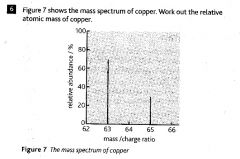
|
63.6
|
|
|
Describe how a mass spectrometer works (the 6 stages).
|
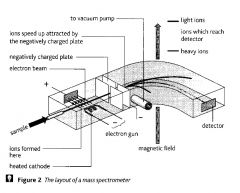
The instrument is kept under a high vacuum so that the ions do not collide with air molecules.
>Vaporisation - sample injected into instrument if gaseous or volatile liquid, if solid is vaporised by heating >Ionisation - sample is passed through stream of high energy electrons from a beam of electrons from electron gun knocks out electrons to form positive ions. Nearly all atoms or molecules lose just one electron and form ions with a 1+ charge but a small number (typically around 5%) lose two electrons to form ions with a 2 + charge >Acceleration - positive ions are attracted towards negatively charged plates and are accelerated to high speed. >Deflection - Magnetic field deflects beam into an arc circle. Heavier ions are deflected less and 2+ ions are deflected twice as much as 1+ ions with same mass. Stronger field, greater deflection. >Detection - magnetic field is gradually increased so that ions of increasing mass enter detector one after another. Ions strike detector, and create a current which is proportional to the abundance of each ion. From strength of magnetic field at which a particular ion hits detector, a computer works out value of charge ratio of original ion. |
|
|
Write down the sub-shells in order of increasing energy up to 4f?
|
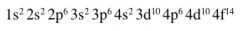
|
|
|
What does arrow-in-box notation look like?
|

|
|
|
a) Which are the two anomalies involving electronic structure?
b) Why? |
a) Chromium (Cr) and Copper (Cu)
b) They donate one of their 4s electrons to the 3d sub-shell (they only have one 4s electron) |
|
|
Define the term first ionisation energy (IE).
|
The energy required to remove one mole of electrons from one mole of gaseous atoms to form one mole of gaseous positive ions.
|
|
|
Sketch a graph of the successive ionisation energies of sodium?
|
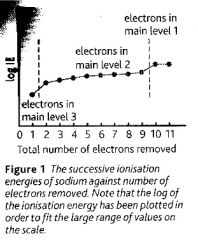
|
|

Sometimes you may need to measure with ruler.
|
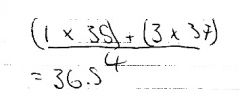
|
|
|
Label axis for spectrometer graph.
|
Side - Abundance (%)
Bottom - Mass:Charge ratio (m/z) |

