![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
7 Cards in this Set
- Front
- Back
|
Three forms of atomic spectroscopy are based on absorption, emission, and uorescence. How do they differ? |
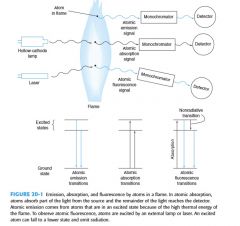
|
|
|
State the advantages and disadvantages of a furnace compared with a ame in atomic absorption spectroscopy. |

|
|
|
State the advantages and disadvantages of the inductively coupled plasma compared with a ame in atomic spectroscopy. |

|
|
|
20-6. Explain how the following background correction techniques work: (a) beam chopping; (b) deuterium lamp; (c) Zeeman. |

|
|
|
20-7. Explain what is meant by spectral, chemical, ionization, and isobaric interference. |

|
|
|
What is the memory effect? |
Interference from previous runs when using furnace. |
|
|
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modier prevents loss of signal from the atom X that readily forms the molecular carbide XC in a graphite furnace (a source of carbon). For example, adding yttrium to a sample containing barium increases the Ba signal by 30%. The bond dissociation energy of YC is greater than that of BaC. Explain what is happening to increase the Ba signal. |

|

