![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
149 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Size of cells |
Virus - 100 nm (1 billionth of a meter) Bacteria - 1 to 10 um (1 millionth of a meter) Eurakyotic cells - 10 to 100 um (Nanometer/micrometer) |
|
|
|
Why are cells not bigger in an elephant? |
Cells are limited by surface area to cell volume ratio. (Volume increases at a higher rate than surface area. Beyond a certain volume the cell does not have enough surface area to import and export all the materials) |
|
|
|
Eukaryotes vs Prokaryotes |
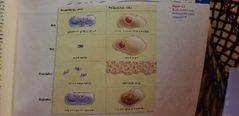
DNA in nucleoid region VS in the nucleus. SIZE smaller VS larger ORGANIZATION usually single VS multicellular OXYGEN not always VS most always ORGANELLES none VS plenty |
|
|
|
Nucleus - EUKARYOTIC CELLS |
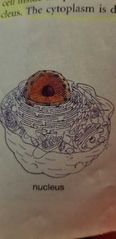
Storage of heredity info, control of cell |
|
|
|
Cytosol - EUKARYOTIC CELLS |

Jelly like fluid inside cells |
|
|
|
Cytoskeleton - EUKARYOTIC CELLS |
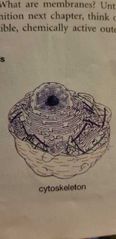
Cellular structure, transport and cellular movement Celia/flagella - current and locomotion |
|
|
|
Plasma membrane - EUKARYOTIC CELLS |

Boundary of the cell, exchange with the environment. |
|
|
|
Nucleolus - EUKARYOTIC CELLS |

Ribosome synthesis |
|
|
|
Nuclear envelope - EUKARYOTIC CELLS |
Exchange between Nucleus and Cytoplasm |
|
|
|
Cytoplasm - EUKARYOTIC CELLS |
The region of the cell inside the plasma membrane but outside the nucleus (incl. organelles) |
|
|
|
Rough Endoplasmic Reticulum (RER) - EUKARYOTIC CELLS |

Protein processing Protein processing |
|
|
|
Endoplasmic Reticulum - EUKARYOTIC CELLS |
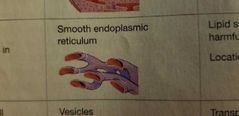
Synthesis of proteins/lipids/steroids Detoxification |
|
|
|
Ribosomes - EUKARYOTIC CELLS |

Site of protein synthesis (free standing in the cytoplasm) Bring the chains of amino acids to the RER for protein synthesis. |
|
|
|
Golgi Complex - EUKARYOTIC CELLS |

Processing, sorting and packaging of proteins. |
|
|
|
Lysosome - EUKARYOTIC CELLS (animal cells only) |
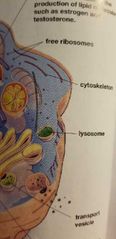
Digestion of (defective) imports, and waste removal.
(Recycling parts or throwing out of the cell) |
|
|
|
Mitochondria - EUKARYOTIC CELLS |

Transform energy from food (Power House). Creates ATP from the respiration of glucose |
|
|
|
Vesicles - EUKARYOTIC CELLS |

Storage and transport of material |
|
|
|
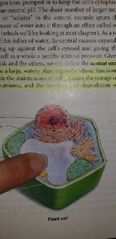
Chloroplasts - EUKARYOTIC CELLS (Plants only) |
Conversion of light into energy (photosynthesis) |
Power house in plants |
|
|
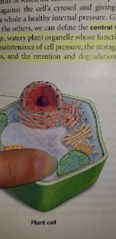
Central vacuole - EUKARYOTIC CELLS (Plants only) |
Storage and excretion. |
The watery organelle |
|
|
Cell wall - EUKARYOTIC CELLS (Plants only) |

Protection Support Fluid pressure |
Only plants have a wall, it makes them static |
|
|
Plasmodesmata - EUKARYOTIC CELLS (Plants only) |
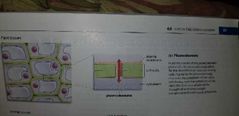
Cell to cell communication |
|
|
|
Gap junction - EUKARYOTIC CELLS (Animal only) |

Cell to cell communication |
|
|
|
mRNA - messenger RNA |

DNA contains information for the making of proteins, the mRNA chains are transmitting the information |
|
|
|
Endomembrane system - EUKARYOTIC CELLS |

The SYSTEM that allow the transport of proteins and other cellular material through the cell.
Starts in the nucleus (and nucleolus), goes by the nuclear membrane pores to the cytoplasma to find the ribosomes, to attach to the RER, then go through a vesicle to the Golgi complex to go through another vesicle to get out of the plasma membrane. |

|
|

To know |
1: cell, cells, cell. 2: eukaryotic, nucleus, prokaryotic. 3: plasma membrane, nucleus, cytosol, organelles. |
|
|

To know |
1: DNA, proteins. 2: ribosomes, RER rough endoplasmic reticulum, Golgi complex. 3: transport vesicle, endomembrane system. |
|
|

To know |
1: smooth ER (endoplasmic reticulum), lysosomes. 2: energy, ATP 3: cytoskeleton |
|
|

To know |
1: chloroplasts, central vacuole, cell wall. 2: lysosome. 3: plasmodesmata, gap junctions. |
|
|
|
Science definition |
A body of knowledge about the natural world. |
|
|
|
Theory definition |
General set of principal, supported by evidences that explains some aspect of nature. |
|
|
|
Fact definition |
Isolated piece of information. |
|
|
|
Scientific method |
Observation Question Hypothesis Experiment Conclusion |
|
|
|
Hypothesis - Definition |
A testable explanation for an observed phenomenon |
|
|
|
Controled condition (in a scientific method) |
An experimental condition that exists prior to the introduction of any variables. |
|
|

|
guess principles nature |
|
|

|
2: hypothesis 3: constant, variable |
|
|
|
Falsifiable - Definition |
Open to negation through scientific inquiry. |
|
|

|
1: revision, evidence 2: negation 3: natural, supernatural |
|
|
|
Biology - Definition |
Study of life |
|
|
|
Characteristics of living things. |
- assimilate and use energy - respond to their environment - maintain a relatively constant internal environement (Homeostasis) - possess DNA that allow them to function - reproduce - composed of one or more cells - evolved from living things - highly organized |
8 |
|
|
Life hierarchy |
Atom> molecule> organelle> cell> tissue> organ> organ system> organism> population> community> ecosystem> biosphere. |
12 |
|
|
Population - Definition |
Member of a single type of living thing (a species) living together in a define area. |
|
|
|
Community - Definition |
Mix of all living things in a given area (forest) |
Forest |
|
|
Ecosystem - Definition |
interaction of non-living elements like water with living members of a community. (California coast) |
California coast. |
|
|
Life science - Definition |
Set of discipline that focus on varying aspects of the living world. |
|
|
|
Evolution - Definition |
Gradual modification of population of living things, resulting sometimes in new species. (Chief principle of "Living thing") |
|
|

|
1: (2,5,1,3,6,4) 2: evolution, species 3: life |
|
|
|
Matter in chemistry - Definition |
Can be defines as anything that takes up space and has a mass. |
|
|
|
Mass - Definition |
Measure of the quantity of matter in a given object. (Matter is space and density) |
|
|
|
Protons, neutrons, electrons |
Protons, positive charge. Defining the atomic number. Neutrons are neutral and often have the same number as protons (unless when they are an isotope) Electrons a negative and are the one who bonds into other substances. Proton and neutron are in the nucleus and represents almost all the mass of the atom. |
|
|
|
Element - Definition |
Substance that is pure and can't be reduce to any simpler component. (Atoms can't be considered as simpler component because they cannot exist by themselves as matter). |
|
|
|
Isotopes - Definition |
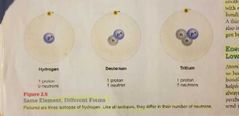
The number of neutrons a nucleus has. (Same element, different form) |
|
|

To know - Chemistry |
1: proton, neutron, electron 2: element, proton 3: neutrons |
|
|
|
Chemical bonds |
Covalent (polar & non-polar) Ionic Hydrogen (They want to be more stable, fill their outer shell) |
3 |
|
|
Covalent bond - Definition |
Share a pair of electron (Water 1 oxygen + 2 hydrogen) |
|
|
|
Ionic bond - Definition |
Occurs when electrons are lost or gain because one atom has a much greater electronegativity. Ionic compound = ions electrostatic attract to each other |
|
|
|
Ions - Definition |
Charged atom after losing or gaining electrons. Become positive when their number of protons is more than their number of electron. Become a negative ion when the atom gained electrons from another atom, making its number of proton lesser than its number of electrons. |
|
|
|
Hydrogen bond - Definition |

A partial negative and partial positive atom attracted to each other. Usually hydrogen with oxigen or nitrogen.
(Water has a hydrogen bond) |
Water |
|
|
Polar vs non polar (covalent bond) |
Polar happens when a molecule takes on a polarity, or a difference in electric charge at one end as opposed to the other. Non polar has symmetric charges. (Water is polar, oxygen attracts the electrons more often around its atom than the hydrogens atoms, oxygen is more electronegative and thus has a more negative charge) |
|
|
|
Molecule - Definition |
An entity consisting of a defined number of atoms covalently bonded together. |
|
|
|
Law of conservation of mass |
Matter is neither created nor destroyed in a chemical reaction. |
|
|

To know |
1: filled, 8, 2 2: covalent 3: polar, electrical charge |
|
|
|
Free radical- Chemistry |

Free radial are important because they shape the molecules. |
|
|
|
Molecular formula Structural formula Ball-and-stick model Space-filling model |
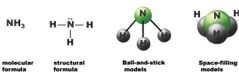
|
|
|
|
Shape in molecule |
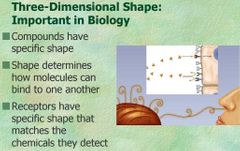
Shapes in molecule are important because they only bind with the appropriate shape receptor |
|
|

To know - Chemistry |
1: loses, electrons, charges 2: electronegative, oxygen, nitrogen 3: bind |
|
|
|
Solution, solute, solvent - Water |
Homogenous mixture being dissolved doing the dissolving |
|
|
|
Water properties |
- Water freezes and create ice which regulated ocean temperature for fishes. - Specific heat: amount of energy required to raise the temperature of a substance by 1°C. Earth buffer. - Water cohesion: tendency for water molecules to stay together. Cohesion impars surface tension. Water molecules are attracted to other water molecules. |
3 |
|
|
Hydrophilic / hydrophobic |
Love / hate water (Nonpolar covalent molecules are hydrophobic.) It helps to compartmentalize. |
|
|
|
pH scale |
The degree to which something is acidic (a scale). |
|
|
|
Acidic, neutral, basic pH |

Acidic> 0-6.9 Neutral> 7 Basic> 7.1-14 Basic is also called = Alkaline |
|
|
|
Hydroxide ion |
Compounds (if numerous) are strongly basic and can be used to move a solution from acidic toward basic. |
Water (acidic/basic) |
|
|
Buffering systems |
Physiological system that function to keep pH withing normal limits. |
|
|

To know - Water |
1: solute, solvent, solution 2: hydrogen bonds, cohesion, specific heat 3: hydrophobic, hydrophilic |
|
|

To know water |
1: yields, ions, accepts 2: 7, 0, 14 3: neutral 7 |
|
|
|
Bio-molecules |
- Carbohydrates - Lipids - Proteins - Nucleic acids (Contains carbon) |
4 |
|
|
Oganic chemistry - Definition |
A branch of chemistry devoted to the study of molecules with carbon as their central element. |
|
|
|
Functional group - Definition |
A group of atoms that confers a specific property to the biomolecule(carbon-based molecule)
(Carboxyl, hydroxal, amino, phosphate.) |
|
|

To know - Biomolecules |
1: carbon, bonds with other elements 2: atoms, carbon based |
|
|
|
Monomers / polymers |
Carbohydrates: monosaccharides / (disaccharide,) / polysaccharide
Lipids: N/A
Proteins: amino acid / polypetide (or protein simply)
Nucleic acids: nucleotide / nucleic acid (DNA, RNA) |
|
|
|
Carbohydrates monomers |
Energy source Simple sugar = monomer = -ose Always has Carbon, Oxygen and Hydrogen |
|
|
|
Carbohydrates polymers |

Starch, energy for plants. Glycogen, energy in animal liver. Cellulose, structure of plants. Chitin, stucture or skin of arthropods. |
|
|

To know - Biomolecules |
Monomers, polymers |
|
|
|
To know - Biomolecules |

2: carbon, oxygen, hydrogen, monosaccharide, polysaccharides. 3: starch, glycogen, cellulose, chitin |
|
|
|
Lipids |
- Lot of Hydrogen - don't mix with water - (no monomer or polymers) - store energy, compartementalization and insulation. |
|
|
|
Types of lipids |
- oils - fats - hormones - phosolipids (Steroids: cholesterol, estrogen and testosterone, all lipids)
|
|
|
|
Fatty acids (lipids) |
Molecules composed of hydrocarbon and a carboxyl group.
- Saturated fatty acid: no double bonds. - Monounsaturated fatty acid: one double bond. - Polyunsaturated: 2 or more double bonds.
The more bonds the healthier. Removing bonds (hydrogenation) from polyunsaturated is super unhealthy and get hard in room temperature.
Triglyceride: 1 glycerol + 3 fatty acids |
3 types. Food etiquettes |
|
|
Fats storage and use |
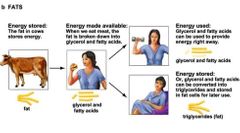
|
|
|

To know - Biomolecules |
1: readily dissolve, water 2: fatty acids, glycerol, carbons atoms, hydrocarbon 3: carbon rings, testosterone and estrogen, phospholipids, phosphate |
|
|
|
Protein |
Series of amino acids that form a chain of protein - Polypeptide
20 amino acids only.
Shape is critical
Always an amino group+ carboxyl group |
|
|
|
Structure in proteins - polypeptides |
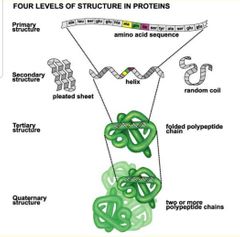
Primary structure: sequence of amino acids.
Secondary: Alpha helix, Beta pleated sheet, random coil.
Tertiary: folded polypetide
Quaternary: 2 or more polypeptide chains. |
4 structures |
|
|
Type of proteins |
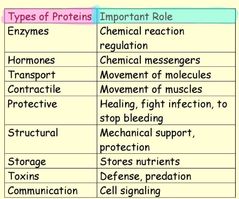
Enzymes, hormones... |
|
|
|
Nucleic acids |
Genetic heredity material Building blocks : nucleotides |
|
|
|
2 type of nucleis acids |
DNA, RNA |
|
|
|
DNA |
Deoxy-ribo-nucleic acid Primary information bearing of life, composed of 2 nucleotides. |
|
|
|
RNA |
Ribo-nucleic acid. Composed of nucleotides. Active in the synthesis of proteins. |
|
|
|
Polymer of DNA |
Phosphate group+ sugar+ nitrogenous base. |
|
|

To know - Biomolecules |
Amino acids, amino acids (To be continued) |
|
|

To know - Biomolecules |
1: Folded up 2: lipoproteins, lipids and proteins 3: nucleotides, phosphate group, sugar, nitrogenous base |
|
|
|
Plasma membrane components |
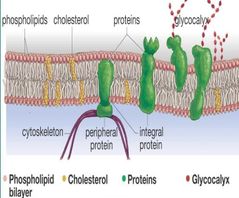
- phospholipid bilayer - cholesterol - proteins - glycocalyx Fluid-mosaic model (mosaic of proteins) |
4 |
|
|
Phospholipid bilayer |
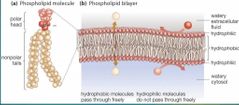
A double layer of phospholipids molecules whose hydrophilic "head" face outward and whose hydrophobic "tails" point inward. Fluid |
|
|
|
Protein in plasma membrane functions |
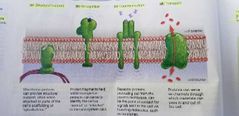
- Structural support - recognition - communication - transport |
|
|
|
Integral protein in plasma membrane |
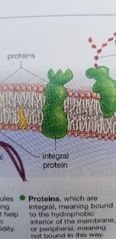
Integral: bound to the hydrophobic interior of the membrane. |
|
|
|
Peripheral proteins in plasma membrane |
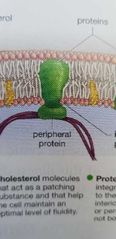
Peripheral: is on either side of the membrane (not bound to its hydrophobic interior). |
|
|
|
Receptor protein - in plasma membrane |
Permit long distance communication through chemical messengers, hormones. Receptor protein binds with signaling molecule. |
|
|
|
Cholesterol in plasma membrane |
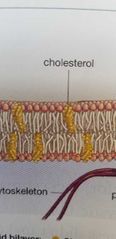
Patching material |
|
|
|
Glycocalyx - in plasma membrane |
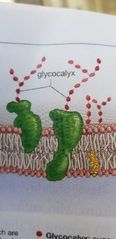
Sugar chain that attach to proteins and phospholipids serving as binding sites, lubrification and adhesion. |
|
|

To know - Plasma membrane |
1: phospholipids bilayer, cholesterol, proteins, glycocalyx. 2: hydrophilic, water, hydrophobic, water. 3: receptor, transport proteins |
|
|
|
Active transport - Plasma membrane (DEFINITION) |
Movement of molecules or ions accross the cell membrane that requires ENERGY |
|
|
|
Passive Transport - Plasma membrane (DEFINITION) |
Movement of molecule or ions accross cell membrane that does NOT require energy. |
|
|
|
Passive transport chanels |
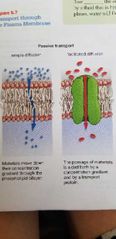
- Diffusion - Osmosis - Facilitated diffusion |
3 passive ways to move material in the cell |
|
|
Diffusion - Definition |

Movement of molecule or ions from a region of higher concentration to lower concentration. |
|
|
|
Osmosis - definition |

The movement of water accross a semipermeable membrane from an area of lower concentration to higher concentration. |
|
|
|
Facilitated diffusion |
Passage of material through the plasma membrane that is aided by a transport protein. |
|
|
|
Concentration gradient - definition |
The difference between the highest and lower concentration of a solute. |
|
|
|
Osmosis in cells |
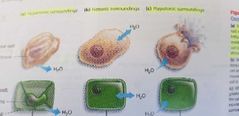
Hypertonic solution: higher concentration of solute in a solution than another. Isotonic: two solution have an equal concentration of solute. Hypotonic: lower concentration of solute in a solution than another |
Hypertonic/isotonic/hypotonic |
|
|
Active transport chanels |
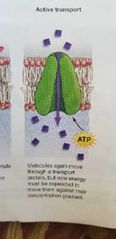
Moves through a transport protein and requires energy source ATP. |
|
|
|
ATP - definition |
Adenosine Triphosphate. Sodium-potasium pump.
Moves larger substance in and out of the plasma membrane. |
|
|
|
Movement of large material through plasma membrane |
-Exocytosis -Endocytosis |
|
|
|
Exocytosis - definition |
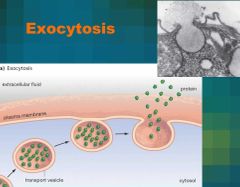
Movement OUT of the cell by fusion. |
|
|
|
Endocytosis - definition |
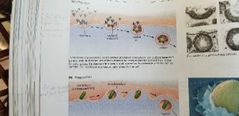
Movement IN the cell.
- pinocytosis (material get detached from the membrane to a vesicle) drinks it, sucks it in.
- phagocytosis (material get surrounded and fuse before to be detached) Eats it, bites it in. |
|
|

To know - plasma membrane |
1: higher concentration, lower concentration, energy. 2: semipermeable, solute concentration, solute concentration. 3: out, in. |
|
|

To know- membrane |
1: active transport, passive transport, protein channel, protein channel. |
|
|

To know- membrane 2: active transport allows for the net movement of molecules into or out of a cell even when there is a... working against this movement. |
2: concentration gradient 3: transport vesicle, plasma membrane, phagocytosis, pinocytosis. |
|
|
|
Energy - definition |
The capacity to bring about movement against an opposite force. |
|
|
|
Potential energy vs kinetic |
Stored energy vs energy in motion |
|
|
|
Thermodynamics |
Study of energy |
|
|
|
1st law of thermodynamics |
Energy is never created of destroyed but is only transformed. |
|
|
|
2nd law of thermodynamics |
Energy transfer always result in a greater amount of disorder. (Piece of coal burns into smoke, coal is condensed and organized that smoke is not) |
|
|
|
Type of work |
Mechanical- muscle contraction Transport- movement of molecule against their concentration gradient. Synthetic- simple molecule to complex molecules (amino acids to proteins) |
|
|
|
Exergonic vs endergonic reactions |

|
|
|

To know - energy |
1: potential, kinetic 2: created, destroyed, transformed, disorder 3: heat |
|
|
|
Coupled reaction |
A chemical reaction in which an exergonic reaction powers an endergonic reaction. |
|
|
|
ATP/ADP cycle |
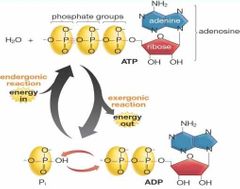
When a ATP (Triphosphate) loses a phosphate group in an exergonic reaction it becomes ADP (Diphosphate). When ADP gain a phosphate group in an endergonic reaction it becomes ATP. |
|
|

To know - energy |
1: product, reactant, reactant, product. |
|
|

To know - energy |
2: ATP, phosphate group 3: ADP, phosphate group |
|
|
|
Enzyme - definition |
A type of protein that accelerates a chemical reaction. (Quicken the metabolic pathway) |
|
|
|
Metabolism - energy |
The sum of all the chemical reactions in an organism. |
|
|
|
Metabolic pathway - energy |
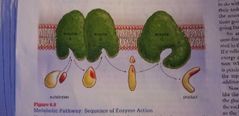
Multi-steps process for enzyme. Each enzyme takes only one task leaving the next task to the next specialized enzyme. |
|
|
|
Substrate - definition |
The substance that is worked on by an enzyme. |
|
|
|
Activation energy - definition |

The energy required to initiate a chemical reaction |
|
|
|
Catalysts - energy |
Substance that retain their original chemical composition while bringing a change to a substract. |
|
|
|
Active site - enzyme |
Portion of the enzyme that binds with a substrate helping it to get transformed. |
|
|
|
Coenzymes - definition |
Molecules (other than amino acids) that facilitate the work of enzymes by binding with them. |
|
|
|
Competitive inhibition - enzymes |
The reduction of the activity of an enzyme because the active site of the enzyme (place where the reaction is fasten) is occupied by another compound. |
|
|
|
Allosteric regulation |
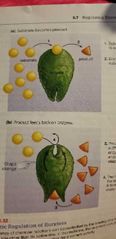
Reduction of the enzyme's activity when a molecule bind to the enzyme in another site than the active site. |
enzymes |
|

To know - enzymes |
1: protein, accelerates 2: substrate, product, substrate 3:initiate, lowering |
|
|
|
Entropy - definition |
A measure of the molecular disorder. (2nd law of thermodynamics) |
|
|
|
Endosymbiosis - definition |
Bacteria that became organelle over time. (Evolution from invader to organelle) |
|

