![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
122 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
What is matter and give examples |
Matter is anything that occupies space and has mass examples are gas liquid and solids |
|
|
|
What is energy and give examples |
Energy is the capacity to do work or to move matter examples of kinetic potential chemical electrical mechanical and radiant |
|
|
|
Which elements make up 96% of living matter |
Carbon hydrogen oxygen and nitrogen |
|
|
|
What are the building blocks of elements |
Atoms |
|
|
|
What is the atomic structure |
Protons electrons and neutrons |
|
|
|
What makes up the mass of an atom |
Protons and neutrons in the nucleus |
|
|
|
Electrons form a negative Cloud around what |
The nucleus |
|
|
|
What is a term that describes the difference in the number of neutrons in the nucleus |
Isotopes |
|
|
|
What happens to the heavier isotopes that are unstable |
They decompose to a stable form by ejecting particles or energy from the nucleus called radioactivity |
|
|
|
What is the smallest unit resulting from The Binding of two or more atoms |
A molecule |
|
|
|
Atoms are said to have a full valence shell when |
Two electrons are in Shell 1 or 8 and the subsequent shells this makes them chemically inactive |
|
|
|
How do valence shells achieve stability |
By losing gaining or sharing electrons to fill the valence shell |
|
|
|
When are ions formed |
When valence shell electrons are completely transferred from one atom to another |
|
|
|
How is an ionic bond formed |
When the opposite charged ions attract each other |
|
|
|
Covalent bonds involve the sharing of what |
Electron pairs between atoms |
|
|
|
When electrons are shared equally the molecule becomes a |
Nonpolar covalent molecule |
|
|
|
When electrons are not shared equally the molecule becomes a |
Polar covalent molecule |
|
|
|
Polar molecules Orient themselves toward |
Charged particles and other molecules |
|
|
|
What is the maximum number of electron shells in any atom known |
7 |
|
|
|
The more distant electrons are more negatively charged and are likely to |
Interact with other atoms |
|
|
|
What are negatively charged ions called |
Anions |
|
|
|
When an atom loses an electron it becomes a positively charged ion called |
Cation |
|
|
|
A good example of ionic bonding is |
Sodium chloride common table salt |
|
|
|
What is an example of a weak Bond |
Hydrogen bond |
|
|
|
What are three recognizable patterns of chemical reactions |
Synthesis reactions decomposition reactions exchange reactions |
|
|
|
When do synthesis reactions occur |
When two or more atoms or molecules combine to form a larger more complex molecule |
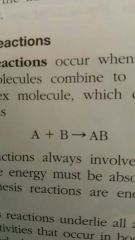
|
|
|
Describe and give an example of what synthesis reaction would look like |

|
|
|
|
Why are synthesis reactions important in the body |
For growth and repair of worn-out or damaged tissues |
|
|
|
When do decomposition reactions occur |
When a molecule is broken down into smaller molecules atoms or ions |

|
|
|
As bonds are broken during decomposition reactions what type of energy is released |
Chemical energy |
|
|
|
Decomposition reactions underlie all what processes that occur in body cells |
Catabolic or destructive processes they are molecule degrading reactions |
|
|
|
Give an example of a decomposition reaction that occurs in the body |
Digestion of foods in the breakdown of glycogen to release glucose |
|
|
|
What exactly is an exchange reaction |
It is the involvement of both synthesis and decomposition reactions bonds are both made and broken |
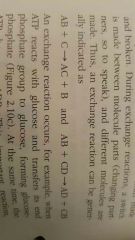
|
|
|
Describe and give an example of what decomposition reactions would look like |

|
|
|
|
Describe and give an example of what an exchange reaction would look like |
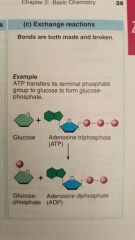
|
|
|
|
What is the difference between inorganic compounds and organic compounds |
Inorganic compounds lack carbon and tend to be small simple molecules such as water and salts organic compounds are carbon containing compounds and examples are carbohydrates lipids proteins and nucleic acids |
|
|
|
Name four roles of water |
They have a high heat capacity they have polarity solvent properties that contain chemical reactivity and they provide cushioning |
|
|
|
When we say water has a high heat capacity what does that mean |
It absorbs and releases large amounts of heat before it's temperature changes and prevent the sudden change in body temperature that might result from intense sun or chilling Winter Winds |
|
|
|
Because of water's polarity it is an excellent solvent it is often called what |
The universal solvent |
|
|
|
What is a solvent |
A liquid or gas in which smaller amounts of other substances called solutes can be dissolved or suspended |
|
|
|
Water is an important reactant in some types of chemical reactions which chemical reactions |
Food digestion breaking down biological molecules |
|
|
|
Describe a cushioning that water serves as a protective function |
In the form of cerebrospinal fluid water forms of Christian around the brain or amniotic fluid within a mother's body |
|
|
|
When dissolved in body fluids salts easily separate into other ions this process is called |
Disassociation because the ions have already been formed |
|
|
|
Define an acid |
A substance that can release hydrogen ions in detectable amounts |
|
|
|
We also call acids |
Proton donors |
|
|
|
The release of what determines an acids effects on the environment |
The release of protons |
|
|
|
In an ionic form what is involved in nerve transmission muscle contraction blood clotting transport of oxygen by hemoglobin metabolism and Bone hardness |
Salt |
|
|
|
Acids release what when dissolved in water |
Hydrogen bonds |
|
|
|
The most important inorganic bases are |
Hydroxides |
|
|
|
What happens when bases and acids interact |
Neutralization occurs |
|
|
|
A pH below 7 is called and a pH above 7 is called |
Below seven is acidic above 7 is alkaline |
|
|
|
What do carbohydrates contain |
Carbon hydrogen and oxygen in the relationship of two hydrogen atoms to one oxygen atom and one carbon atom |
|
|
|
What are the building blocks of carbohydrates |
Monosaccharides |
|
|
|
What is the major source for the formation of ATP |
Carbohydrates in particular glucose |
|
|
|
Give examples of some lipids |
Triglycerides phospholipids steroids cholesterol |
|
|
|
How are lipids degraded and synthesized |
They are degraded by hydrolysis in synthesized by dehydration synthesis |
|
|
|
How are proteins constructed |
From the building blocks called amino acids |
|
|
|
Name two nucleic acids |
Deoxyribonucleic acid and ribonucleic acid |
|
|
|
What is the building unit of nucleic acid |
Nucleotide |
|
|
|
What is the role of DNA |
Maintains genetic Heritage by replicating itself before cell division and specifying protein structure |
|
|
|
What is the role of RNA |
Attacks in protein synthesis to ensure that instructions of DNA are executed |
|
|
|
What does ATP stand for |
Adenosine triphosphate |
|
|
|
What is ATP |
It is the universal energy composed usually by cells of the body |
|
|
|
What is a cell |
Cells are the structural units of all living things |
|
|
|
Primarily what are the elements that make up a cell |
Carbon oxygen hydrogen and nitrogen |
|
|
|
What are the three main regions that make up a cell |
The nucleus , cytoplasm ,plasma membrane |
|
|
|
The control center of the cell is called the |
Nucleus |
|
|
|
There are three recognizable regions or structures that the nucleus has and they are called |
Nuclear envelope nucleoli and chromatin |
|
|
|
The nucleus is bounded by a double membrane barrier called |
Nuclear envelope or nuclear membrane |
|
|
|
The nuclear membrane encloses a jelly-like fluid called |
Nucleoplasm |
|
|
|
The nucleus contains one or more small dark staining round bodies called |
Nucleoli |
|
|
|
Explain what happens in the nucleoli |
Ribosomes are assembled here |
|
|
|
Where do ribosomes serve as the actual sites of protein synthesis |
Cytoplasm |
|
|
|
When a cell is not dividing its DNA is combined with protein and forms a loose network of bumpy threads called |
Chromatin |
|
|
|
When a cell is dividing to form two daughter cells the chromatin threads coil and condense to form dense Rod like bodies called |
Chromosomes |
|
|
|
What is the plasma membrane |
A fragile transparent barrier that contains the cell contents and separates them from the surrounding environment |
|
|
|
Describe the structure of the plasma membrane |
It consists of two lipid or fat layers arranged tail to tail in which protein molecules float the proteins are free to move and form a constantly changing pattern or mosaic |
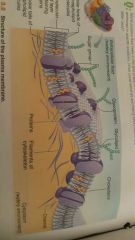
|
|
|
Which part of the phospholipid molecules are hydrophilic and which part are hydrophobic |
The heads of the molecules are hydrophilic and the tails are nonpolar and hydrophobic |
|
|
|
What are some of the important features of glycoproteins |
determine your blood type act as receptors that certain bacteria viruses or toxins can bind to and play a role in cell to cell recognition and interactions |
|
|
|
Describe three ways that cells can be bound together |
Glycoproteins in the glycocalyx act as an adhesive or wavy Contours of the membranes of cells fit together in a tongue and groove fashion or special membrane Junctions are formed |
|
|
|
What are tight junctions |
Impermeable Junctions that binds cells together into leak-proof sheets that prevent substances from passing through |
|
|
|
What type of Junctions are desmosomes |
In Grand Junction scattered like rabbits along with sides of cells |
|
|
|
In gap Junctions the neighboring cells are connected by a hollow cylinders composed of proteins called |
Connexons |
|
|
|
What is the cytoplasm |
The cellular material outside the nucleus and inside the plasma membrane |
|
|
|
The cytoplasm includes three major elements what are they |
Cytosol organelles and inclusions |
|
|
|
What does the cytosol of the cytoplasm do |
It is a semi-transparent fluid that suspends the other elements |
|
|
|
What do the organelles of the cytoplasm do |
They are the metabolic Machinery of the cell that carries out a specific function for the cell |
|
|
|
What are the inclusions of the cytoplasm and what do they do |
Inclusions are chemical substances that may or may not be present depending on the specific cell type they are stored nutrients or cell products |
|
|
|
What are the powerhouses of the cell have a constant supply of ATP |
Mitochondria |
|
|
|
What are the actual sites of protein synthesis in the cell |
Ribosomes |
|
|
|
What is the name for the whole ribosome membrane combination |
Rough endoplasmic reticulum |
|
|
|
What accounts for about half of a cell's membranes and is a system of fluid-filled cisterns that coil and twist throughout the cytoplasm |
Endoplasmic reticulum |
|
|
|
What is the difference between rough endoplasmic reticulum and smooth endoplasmic reticulum |
Rough endoplasmic reticulum is important in cells that make an export proteins for pancreas cells digestive enzymes in the small intestine smooth endoplasmic reticulum plays no protein and protein synthesis and functions in lipid metabolism |
|
|
|
Describe the appearance of the Golgi apparatus |
A stack of flattened membranous Saks associated with swarms of tiny vesicles |
|
|
|
What is the purpose of the Golgi apparatus |
To modify and package proteins in specific ways |
|
|
|
What happens after proteins are ready for export and they accumulate in the Golgi apparatus |
This Axwell and they're swollen and fill with protein pinch off and form secretory vesicles which travel to the plasma membrane |
|
|
|
What are lysosomes |
Membranous bags containing powerful digestive enzymes |
|
|
|
What membranous sacs containing powerful oxidase enzymes that use molecular oxygen to detoxify a number of harmful or poisonous substance |
Peroxisomes |
|
|
|
Name the internal framework that determines a cell's shape supports other organelles and provides the machinery for intracellular transport |
Cytoskeleton |
|
|
|
Describe centrioles |
They are made up of fine microtubules in a rod-shaped and they generate during cell division they direct the formation of the mitotic spindle |
|
|
|
What is the role of cilia |
To move substances along the cell surface |
|
|
|
Projections formed by the centrioles or substantially longer they are called |
Flagella |
|
|
|
What is the difference between a cilia and flagella |
Cilia Propel other substances across the cell surface whereas flagella propels the cell itself |
|
|
|
Microvilli are |
Finger-like extensions of the plasma membrane that project from an exposed cell surface |
|
|
|
The elongated shape of this cell lies along the cable like fibers that it secretes it has in abundance of rough ER in a large Golgi apparatus to make and secrete the protein building blocks of these fibers what is this cell |
Fibroblast |
|
|
|
This cell carries oxygen in the bloodstream its concave disk shape provides extra surface area for the uptake of oxygen and streamlines so that this cell can easily flow through the bloodstream name this cell |
Erythrocyte |
|
|
|
The cell is a hexagon shape and it covers and lines body organs which cell is this |
Epithelial cell |
|
|
|
These cells are filled with abundance contractile filaments so they can shorten forcefully and move the bones or change the size of internal organs name the cells |
Skeletal muscle and smooth muscle cells |
|
|
|
The huge spherical shape is produced by a large lipid droplet in its cytoplasm name this cell |
Fat cell |
|
|
|
The cell extends along pseudopods or false feet to crawl through tissue to Reach Infection sites name this cell |
Macrophage |
|
|
|
What is the difference between intracellular fluid and interstitial fluid |
Intracellular fluid is a solution containing small amounts of gases nutrients and salts dissolved in water interstitial fluid continually based exterior of our cells |
|
|
|
Substances move through the plasma membrane in what two ways |
Passive or active |
|
|
|
Name two passive processes |
Diffusion and filtration |
|
|
|
Describe diffusion |
Diffusion is when molecules move from an area of concentration to an area of less concentration |
|
|
|
Give examples of four different types of diffusion |

|
|
|
|
The process by which water and solute are forced through a membrane by fluid or hydrostatic pressure is called |
Filtration |
|
|
|
Whenever a cell uses some of its ATP Supply to move substances across the membrane the process is referred to as |
Active |
|
|
|
What is active transport |
A process that requires protein carriers combined with the substances that are to be transported across the membrane |
|
|
|
What type of transport helps from ATP and moves substances into or out of cells without their actually crossing the plasma membrane |
Vesicular transport |
|
|
|
What is exocytosis |
Hit move substances out of cells cells actively secrete hormones mucus and other cell products or reject certain cellular wastes |
|
|
|
What is endocytosis |
ATP requiring process that takes up and go ventricular substances by closing them in a small membranous vesicle |
|
|
|
What is the difference between phagocytosis and pinocytosis |
Phagocytosis is cell eating and pinocytosis is cell drinking |
|

