![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
104 Cards in this Set
- Front
- Back
|
List roles of cell membrane |
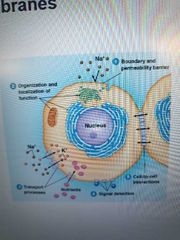
1. Define cell and organelle boundaries as permeability barriers
2. Sites for biological funct like electron transport
3. Contain transport proteins that move stuff in/out of cells/organelles
4. Detects external signals via its protein molecules that act as receptors
5. Provide mechanism for intercellular contact, adhesion, communication
|
|
|
What makes the cell membrane act as permeability barriers? |
Hydrophobic interior |
|
|
Plasma vs intracellular membrane? |
Plasma - around whole cell Intracellular- compartmentalize functions in eukaryote cell |
|
|
How do membranes perform specific functions and how are they recognized? |
Molecules (mostly proteins) responsible for functions are embedded in or bound to membranes. These molecules (enzymes) are used to identify/specialize a membrane |
|
|
What enters and leaves the cell? How? |
Enter - nutrients, ions, gas, h2o, etc Leave - waste and products How -direct diffusion (small, nonpolar molecules) or via transporters |
|
|
What is signal transduction? |
Mechanism for how electric or chemical signals from the environment transmit to cell interior |
|
|
How do membrane receptors work? |
Chemical signal molecules bind to membrane protein receptors on the outer surface of the membrane. This signals chemical events on the inner membrane, which leads to change in cell function |
|
|
What are cadherins? |
Membrane proteins with extracellular amino acid sequences that bind Ca and promote adhesion between similar cell types in a tissue |
|
|
List types of intercell junctions besides cadherins |
Animal tissue: Adhesive junctions -hold cells together Tight junctions- makes seal to block passing of fluid between cells Gap junctions- "telephone" that allows communication between neighboring cells Plants: Plasmodesmata -see gap junctions |
|
|
What is the fluid mosaic model? |
2 layers of fluid, mobile lipids with a protein mosaic within and on top |
|
|
Describe Overton and Langmuir's discoveries |
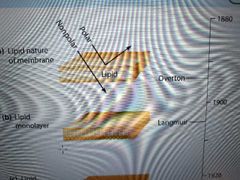
Overton - cell surface must have a lipid coat because he saw nonpolar, lipid-soluble substances easily entered cells
Langmuir - discovered phospholipids were amphipathic, oriented on water with hydrophobic tails away from water (not a bilayer, yet). He dissolved lipids in benzene and layered the sample on water, where it formed a 1-molecule thick monolayer. |
|
|
Describe Gorter and Grendel's discoveries |

Extracted erythrocyte lipids and layered them on water using Langmuir's method. Lipid film was 2x surface area of blood cells, suggesting a bilayer with hydrophobic regions inward |
|
|
What did Davson and Danielli discover? |
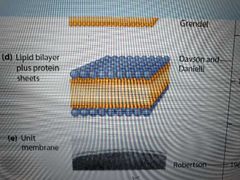
Suggested that a protein sheet coated the lipids as an explanation for a membrane's:
Electrical resistance Solute permeability Surface tension (surface tension of a created liquid film was much higher than a natural cell membrane's, and could be lowered by adding protein) |
|
|
What did Robertson discover? |

Labelled cell membrane with osmium and sliced it open to see inside using transmission electron microscope Saw that all membranes share a unit membrane trilaminar structure |
|
|
What ideas challenged the Davson-Danielli model? |
Davson-Danielli - protein coat model
Membranes can be digested by phospholipase so lipids must be exposed. Only some areas covered by peripheral proteins were preserved.
Surface proteins could not be isolated without organic solvents or amphipathic detergents such as SDS, meaning they were hydrophobic and NOT coating the layers, but located within the layers |
|
|
What did Unwin and Henderson discover? Example? |
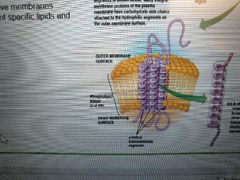
Most integral proteins have 1+ hydrophobic alpha helix transmembrane segments that anchor the protein to the membrane
Ex: bacteriohodopsin, which lets cell use E from sunlight |
|
|
How are membrane lipids ordered? |
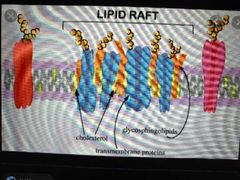
Through lipid raft microdomains.
Lipid travel in groups that all perform a specific function - they do not mix together freely. |
|
|
List classes of membrane lipids |
- Phospholipids (most commonly seen because they make the membrane structure) -> phosphoglycerides -> sphingolipids
Glycolipid -> glycoglycerolipids -> glycosphingolipids *cerebrosides *gangliosides
Sterol -> cholesterol -> phytosterols -> ergosterol |
|
|
How are glycolipids and sterols distinguished? |
Glycolipids have carbohydrate additions Sterols have a hydroxyl group that H-bonds to polar region of membrane (allows sterol to attach to membrane) |
|
|
What are cerebrosides and gangliosides? |
Prominent in brain and nerve cells Cerebroside - neutral glycolipid with uncharged sugar as head group Ganglioside - oligosaccharide head group with 1+ negatively charged sialic acid residues |
|
|
Common types of sterols? |
Animal membrane - cholesterol (stabilize and maintain membrane) Plant - phytosterols Fungi - ergosterol |
|
|
Role and size of fatty acids in membranes? |
Long hydrocarbon tails that make a barrier to polar solute diffusion Average size is 12-18 C's - optimal for bilayer formation and membrane thickness (6-8nm) |
|
|
Saturated vs unsaturated fatty acids with examples |
Saturated - no double bonds. ->palmitate (16C) 16:0 = C:# double bonds ->stearate (18C) 18:0
Unsaturated- 1 double bond -> Oleate (18C) 18:1 Polyunsaturated - 2+ double bonds -> linoleate (18C) 18:2 -> omega-3 (for human development and reduced heart disease risk) |
|
|
What is membrane asymmetry and when is it established? |
different lipids and degrees of saturation of fatty acids in phospholipids found in each monolayer (ex: most glycolipids found in outer layer of animal plasma membranes)
Est. During membrane synthesis |
|
|
List types of lipid movement |
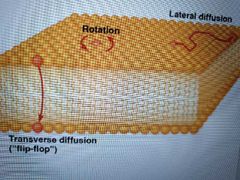
Transverse diffusion- lipid flips from one monolayer to the other. Rarely occurs. Ex: proteins called phospholipid translocators or flippases catalyze flipping in smooth ER membrane Rotation- circles around axis (rapid and random) Lateral diffusion - across monolayer (rapid and random) |
|
|
How fast can lipids move? |
Several micrometers per sec |
|
|
How is speed of lateral diffusion demonstrated? |
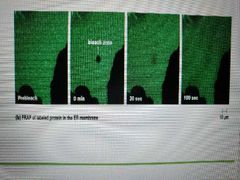
Fluorescence recovery after photobleaching (FRAP) 1. Label lipids with fluorescent dye 2. Laser beam bleaches dye in small area 3. Timer is set to see how long bleached spot disappears as a result of lipid movement |
|
|
What is Tm? |
Transition temperature - T at which bilayer becomes fluid |
|
|
What is a membrane's phase transition? |
Change of membrane from solid to liquid as it reaches Tm
Cold = more solid Hot = more fluid |
|
|
How is Tm experimentally determined? |
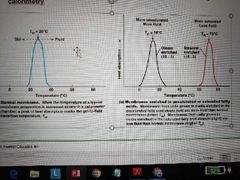
Differential scanning calorimetry Membrane put into cslorimeter and uptake of heat is measured. T at which membrane becomes fluid is point of maximum heat absorption. Fluid membranes will have lower Tm than solid, saturated membranes (less heat to liquify a membrane that is already partially fluid) |
|
|
How does fatty acid composition affect fluidity? |
High Tm/solid: -> longer-chain tails (more C) -> saturated
Low Tm/fluid: -> short chain -> unsaturated (bends from double bonds)
|
|
|
How is membrane fluidity maintained at physiological temps? |
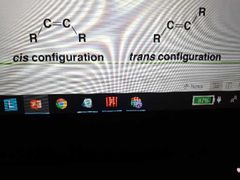
-Most plasma membrane fatty acids vary in length and degree of saturation
-unsaturated fatty acids naturally have cis conformation because they have more of a bend than commercially made trans fats (which pack tighter) which allows more fluidity
|
|
|
How do sterols (cholesterol) affect membrane fluidity? |
Cholesterol is a fluidity buffer that helps maintain an ideal amount of fluidity at all times- insertion into membrane increases Tm and decreases fluidity at high temps, but at cold temps, it prevents phospholipids from packing tightly to solidify the membrane |
|
|
How do sterols affect membrane permeability? |
Decreased permeability to ions and small polar molecules by filling up the space (route) between hydrocarbon chains |
|
|
How do organisms regulate membrane fluidity? Use examples |
1. Vary lipid composition
Ex: poikilotherms cant regulate body temp so they use homeoviscous adaptation to alter length and degree of saturation of membrane fatty acids
2. Desaturase enzyme adds double bonds as needed to increase fluidity
Ex: plants and yeasts - oxygen is more available (increased solubility) at cold temps and can be used as substrate for desaturase |
|
|
What are lipid rafts? |
Localized regions of membrane lipids with specialized cell-signalling proteins, which change composition as lipids/proteins move in and out of them |
|
|
How are animal cell lipid rafts characterized? |
Elevated concentration of cholesterol and glycosphingolipids, which have longer, saturated tails and create tightly packed, less fluid sections of the membrane |
|
|
List 2 types of the 200 raft-associated proteins |
Lipoproteins with fatty acids attached Actin-binding proteins that suggest cytoskeleton has role in raft formation and organization |
|
|
How can proteins be blocked from targeting lipid rafts? |
Depleted cholesterol Disrupted actin cytoskeleton |
|
|
List examples of extracellular signaling roles of lipid rafts |
Transport nutrients and ions across membrane Bind activated immune system cells to microbial targets Transport cholera toxin into intestinal cells |
|
|
What is the relation between lipid rafts and receptor proteins? |
Receptor proteins that detect external chemical signals bind to outer monolayer of plasma membrane and can move into lipid raft located on the same monolayer. The raft it moves to is coupled to another raft on the inner monolayer, some of which have kinases, which catalyze phosphorylation of target molecules inside the cell. This generates chemical signaling inside the cell. |
|
|
List types of membrane proteins |
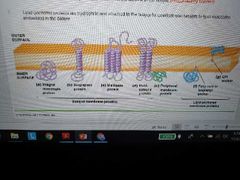
Integral - embedded in bilayer, hydrophobic, can be monotropic (embedded in only one side) or transmembrane
Peripheral - weak electrostatic force and H-bonded, hydrophilic, located on surface with some hydrophobic residues to anchor them
Lipid anchored - covalently bonded to embedded lipid molecules but found on surface, hydrophilic |
|
|
Are most integral proteins monotropic or transmembrane? |
Transmembrane |
|
|
How are transmembrane proteins anchored? Describe structure |
Anchored by 1+ hydrophobic transmembrane segments, mostly alpha helices, but some are B barrel beta sheets (ex: porins) |
|
|
Singlepass vs multipass proteins and examples |
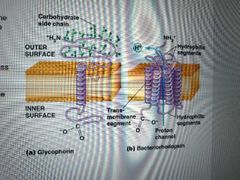
Single -C-terminus extends out of one side and N-terminus out the other Ex: glycophorin Multi - 2+ transmembrane segments Ex: bacteriohodopsin (6 transmembrane segments that form a channel) |
|
|
List types of lipid-anchored membrane proteins |
GPI-anchored - external proteins covalently linked to glycosylphosphatidylinositol
Fatty-acid anchored - internal proteins covalently linked to isoprenyl groups (saturated fatty acids), usually myristic or palmitic acids
|
|
|
How are isoprenylated membrane proteins formed? |
Made in cytosol and modified by adding multiple isoprenyl groups (5c) , usually farnesyl (15c) or geranylgeranyl(20c) groups |
|
|
How are peripheral and lipid anchored proteins isolated? |
Solubilized and extracted by: Altering pH or ionic strength Chelating (cation-binding) agents |
|
|
How does electrophoresis work? |
1. Solubilize membrane fragments in sodium dodecyl sulfate (sds) to disrupt protein-protein and protein-lipid interaction 2. Proteins are now evenly coated with negatively charged detergent molecules 3. Load proteins onto agarose or polyacrimide gel and apply electrical potential 4. Proteins separate by charge and size (small molecules move faster) 5. Polypeptides IDed by Western blotting |
|
|
What does SDS-PAGE stand for? How does 2-d SDS-PAGE separate proteins? |
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis Separates first by charge, then by size |
|
|
What is western blotting? |
Polypeptides from electrophoresis gel are transferred to a nitrocellulose membrane and bound with antibodies that only bind to specific polypeptides |
|
|
What is membrane reconstitution? |
Membrane Proteins are extracted and separated individually, then mixed with phospholipids to form a liposomes/artificial membrane vesicles These false membranes can be loaded with particular molecules to examine their ability to perform specific, targeted functions |
|
|
What is homeostasis? |
Cells accumulate a variety of components in concentrations different from its surroundings |
|
|
List types of transport and which solutes are transported |
Simple diffusion - small polar (ex: h2o, glycerol, ethanol); small and lg nonpolar
Facilitated (passive) diffusion- sm and lg polar, ions
Active transport - lg polar (ex: estrogen, testosterone), ions |
|
|
What are transport proteins? Give example |
Integral proteins that recognize specific substances to be transported across membrane Ex: aquaporin transports only h2o |
|
|
Facilitated diffusion vs active transport? |
Facilitated/passive doesnt require energy because it moves solutes from high to low concentrations (exergonic - ^G movement)
Active moves solutes against concentration gradient (low to high, endergonic + ^G movement) and requires energy by ATP hydrolysis or transporting another solute down its concentration gradient |
|
|
What determines solute movement (diffusion) across a membrane? |
Size, polarity, charge. No charge- concentration gradient
Charge (ions) - electrochemical potential (concentration gradient + charge gradient) |
|
|
What is membrane potential? |
Charge gradient caused by active transport of ions. Symbolized by "Vm" |
|
|
Molecular weight required for simple diffusion? |
Under 100 Ex: ethanol is 46, glycerol is 92. Glucose needs transporter because its weight is 180 |
|
|
What is osmosis? |
A form of simple diffusion. When solutes cant diffuse across membrane, water will diffuse to equalize concentrations |
|
|
What is osmolality? |
Solute concentrations inside vs outside cell |
|
|
Explain Isotonic, hypertonic, hypotonic solutions |
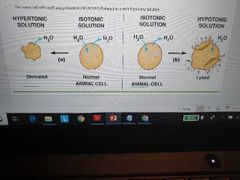
Isotonic - solute concentrations equal in and out of cell
Hyper - solute concentration higher outside cell. Cell Shrivels because water will leave the cell to balance all thensokutes outside of it
Hypo - solute concentrations higher inside cell. Cell bursts/loses because water enters the cell trying to match its solute concentration to the outside
|
|
|
Describe Alec Bangham's discovery |
He trapped solutes inside liposomes and measured their diffusion rates. Ions were trapped for days but small uncharged molecules like h2o diffused too fast to measure |
|
|
Why is water theorized to diffuse faster than expected? |
1. Membranes may have pores that allow water to diffuse rapidly based on polarity 2. Membrane lipid movement creates holes that water can slip through |
|
|
Why dont ions diffuse easily? |
Water forms hydration shells around polar molecules and require energy to break them |
|
|
Why is ion impermeability important to cell activity? |
Each cell membrane needs to maintain an electrochemical potential (concentration gradient + charge gradient), either Na (animal cell) or protons (other cells) But, ions must still be allowed to cross in a controlled manner |
|
|
What is the role of a transport protein in facilitated diffusion? |
All it does is provide a channel through the bilayer for polar molecules to pass along its concentration gradient |
|
|
List types of transport proteins and what they do |
Carrier proteins (transporters and permeases) bind solute on one side, change shape, release solute on other side (this is the alternating conformational model) Channel proteins form hydrophilic channel/tunnel |
|
|
List types of channel proteins |
ion channels - Small, highly selective channels (most only accept 1 type of ion like Na, Ca, Cl, etc) - voltage gated open and close in response to membrane potential (ion gradient caused by active transport) change
- ligand gated open and close due to substrate binding to the protein
- mechanosensitive open and close from mechanical force on the membrane
Aquaporins - water
Porins - Large, nonspecific channels (pores on outer membranes of bacteria, mitochondria, chloroplasts) - pores formed by porin proteins that allow solutes up to 600 molecular weight
|
|
|
Why are ion channels faster than carrier protein transport? |
No conformational change required |
|
|
How are carrier proteins like enzymes? |
- regulated by external factors - diffusion means a substrate binds to a specific site - solute-carrier complex intermediate is formed - product released after conformational change |
|
|
Example of carrier protein's high specificity |
Protein specific for a single compound or small group of closely related compounds Ex: carrier for erythrocyte glucose is specific to a few monosacchararides AND stereospecific for only D isomers |
|
|
Explain saturation kinetics |
Carrier proteins become saturated because they are on limited numbers and reach a finite maximum velocity, however solute concentrations rise above this amount. |
|
|
What is competitive inhibition? Example? |
Incorrect molecules that are structurally related to the correct solute Ex: mannose and galactose monosaccharides can inhibit glucose transport by glucose carrier proteins |
|
|
How many solutes do carrier proteins transport? |
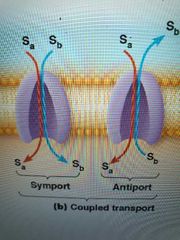
One (uniport) by uniporter or two (coupled transport) by antiporters and symporters - 2 solutes in same direction = symport/co transport - 2 solutes moving opposite directions = antiport/countertransport |
|
|
Describe glucose unitransport |
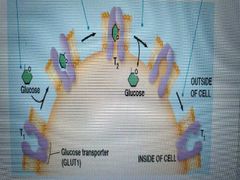
1. D-glucose binds to glut1 transporter in t1 cell conformation 2. Glut1 conforms to t2 cell conformation 3. Glucose is released 4. Glut1 conforms back to t1 cell conformation
|
|
|
Why can erythrocyte uptake glucose by facilitated diffusion and what is glut1 made of? |
Level of blood glucose is much higher than that inside the cell Glut 1 has 12 transmembrane segments and a cavity with hydrophilic sidechains |
|
|
Why is glucose concentration kept low in animal cells? |

When glucose is released into the cell, atp hydrolysis by hexokinase attaches phosphate to make glucose-6-phosphate, used in glycolysis. Also this phosphorylated glucose cant bind to the carrier anymore |
|
|
Why is carrier protein function reversible? |
Proteins can facilitate transport both in and out of cell depending on solute concentrations |
|
|
What is the basis for ion channel selectivity? |
Size Binding sites involving amino acid side chains |
|
|
List ion channel functions |
Cell communication- muscle contraction, nerve cell electrical signaling Salt balance maintenance Airway linking in lungs - cystic fibrosis transmembrane conductance regulator (CFTR) channel maintains Cl concentration in lungs. Defect of protein = cystic fibrosis |
|
|
What is the structure of porins? |
B-barrel transmembrane structure with water-filled pore at center. Polar side chains line the inside, nonpolar line the outside |
|
|
Describe aquaporin structure |
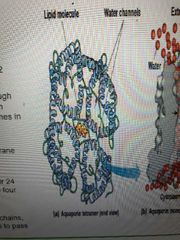
Tetramers with 24 transmembrane segments, forming 4 central channels lined with hydrophilic side chains. Water can pass through these channels 1 at a time |
|
|
Where are aquaporins found? |
Animals - erythrocyte and kidney cells Plants - root and vacuolar membrane cells |
|
|
List functions of active transport |
Uptake of essential nutrients Waste removal Maintenance of non equilibrium levels of certain ions |
|
|
What gives active transport directionality and other transport nondirectionality? |
Active only moves solutes in one direction whereas simple and facilitated diffusion are non directional regarding the membrane and operate based on solute concentration |
|
|
List categories of active transport |
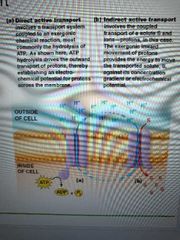
Direct/primary - energy is from exergonic chem reaction (atp hydrolysis). Transporters called ATPases or ATPase pumps
Indirect/secondary - energy is from exergonic transport of one solute down its gradient. This powers the endergonic transport of another solute up (against) its gradient. Can be symport or anti port depending on if both solutes go in the same or opposite directions |
|
|
List types of direct transport ATPases |
P-type V-type F-type Abc-type |
|
|
Function and structure of V-type ATPase |
function - pump protons into organelles (golgi apparatus, vesicles, cajoles, lysosomes, endosomes) Structure - 1 integral multi subunit embedded in the membrane and 1 peripheral multi subunit sticking out of the membrane surface |
|
|
What does ABC-type ATPase stand for? |
ATP Binding Cassette transporter Cassette -the part of the protein that binds to atp |
|
|
Functions of abc-type ATPases |
Importers that uptake nutrients Exporters that pump drugs/antibiotics out of cells, making the cell drug-resistant -drug-resistant tumors have MDR (Multidrug resistance) transport protein |
|
|
List functions of indirect transport proton gradients |
uptake of organic solutes Export of other ions ***pump that maintains electrochemical potential (concentration + charge gradients) is still driven by ATP, but transport is not directly driven by ATP *** |
|
|
List examples of active transport |
Na/K ATPase/pump - direct P-type ATPase transport Na/glucose symporter- indirect transport |
|
|
What is the purpose of the na K pump. What are the na and k ratios? |
Purpose - asymmetric distribution of ions across animal plasma membrane; transmission of nerve impulses
30k inside:1k outside 0.08na inside: 1na outside |
|
|
What drives na/k pump? |
Exergonic hydrolysis of atp drives ion transport |
|
|
Describe structure of na/k ATPase |
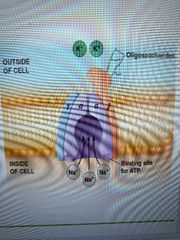
Trimeric transmembrane protein with one a,b,y subunits a has binding sites for K + atp externally and na + atp internally. Nobody knows about b and y functions yet |
|
|
Describe na/k pump's conformational changes |
1. 3 na ions bind to E1 site inside cell 2. ATP phosphorylates alpha subunit 3. Pump conforms to E2 conformation and releases Na outside cell 4. K ions bind to E2 site 5. ATP dephosphorylates alpha subunit 6. Pump conforms to E1, releases K into cell |
|
|
When is a Na/glucose symporter used? |
Most glucose enters and leaves cell by facilitated diffusion but cells lining the intestines use a symporter for glucose and aminos even when concentrations are higher than outside of the cells |
|
|
Describe mechanism of Na symport |
1. 2 external na bind 2. 1 external glucose binds 3. Symporter changes conformation 4. Na enters cell due to low internal na concentration (and pumped back out by na/k pump) 5. Symporter is locked into place until glucose leaves into cell 6. Symporter changes conformation again |
|
|
List p-type ATPases and functions |
P1. All organisms, transport heavy metal ions P2. Eukaryotes, maintains ion gradient(na,k, ca, H) across plasma membrane for muscle contraction and acidification of gastric juice P3. Plants and fungi, pump protons out through plasma membrane to acidify external medium P4. Act as flippase to pump hydrophobic molecules(cholesterol, fatty acid) P5. Cation transport |
|
|
What inhibits p-type atpases? |
Vanadate, because it resembles phosphate |
|
|
Where are f-type atpases found? |
Bacteria Mitochondria and chloroplasts |
|
|
Describe f-type atpase structure |
2 components: F0 - transmembrane pore for protons F1 - peripheral component with ATP binding site |
|
|
What do f-type atpases do? |
Synthesize ATP using E from exergonic flow of protons down their gradient. Found in all eukaryotes, some prokaryotes Instead of atpase, it is an atp synthase ***atp can be used as E for ion gradients and gradients can be used as E for ATP synthesis*** |

