![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
64 Cards in this Set
- Front
- Back
|
Polar molecule |
The opposite ends have opposite charges |
|
|
Four emergent properties of water |
Cohesive behavior, ability to moderate tempurate, expansion upon freezing, versatility as a solvent |
|
|
Cohesion |
When Hydrogen bonds hold water molecules together |
|
|
Adhesion |
An attraction between different substances. Ex. Water and plant cells |
|
|
Surface tension |
The measure of how hard it is to stretch or break the surface of a liquid. It is related to cohesion |
|
|
Moderation of temperature by water |
Water absorbs hear from warmer air and releases stored heat to cool air. Water can absorb or release a large amount of heat with only a slight change in its own temperature. |
|
|
Kinetic energy |
The energy of motion |
|
|
Thermal energy |
The total kinetic energy associated with random movement of atom or molecules (matters volume) |
|
|
Temperature |
The average kinetic energy of the molecules in a body of matter |
|
|
Heat |
The transfer of thermal energy from one body of matter to another |
|
|
Calorie |
The amount of heat required to raise the temperature of 1g of water |
|
|
Joul |
Is another unit of energy where 1J= .239 calories. Or 1cal=4.184J |
|
|
Specific heat |
The amount of heat that must be absorbed or loft for 1g of that substance to change its temperature. |
|
|
Evaporation |
Transformation of a substance from liquid to gas |
|
|
Heat of vaporization |
The heat a liquid must absorb for 1g to be converted to gas |
|
|
Evaporative cooling |
The process of a liquid evaporates it's remaining surface cools |
|
|
Evaporative cooling |
Helps stabilize temperatures in organisms and bodies of water |
|
|
Solution |
A liquid that is homogenous mixture of substances |
|
|
Solvent |
The dissolving agent of a solution |
|
|
Solute |
Substance that is dissolved |
|
|
Aqueous solution |
One in which water is the solvent |
|
|
Hydration shell |
When an ionic compound is dissolved in water each ion is surrounded by a sphere of water molecules called a hydration compound. |
|
|
Hydrophillic |
Substance that has an affinity for water |
|
|
Hydrophobic |
Substance is one that does not have an affinity for water |
|
|
Molecular mass |
The sum of all masses of all atoms in a molecule |
|
|
1mol |
6.02 x 10^23 |
|
|
Avocados number |
6.02 x 10^23 daltons=1g |
|
|
Molarity |
The number of moles of solute per liter of solution |
|
|
Hydrogen (H+) |
The hydrogen atom leaves it's electron behind and is transferred as a proton |
|
|
Hydronium |
The molecule with the extra proton is now a hydronium ion (H+) |
|
|
Hydroxide ion OH- |
The molecule that lost the proton |
|
|
Water in a state of dynamic equilibrium (pic) |

|
|
|
Though statistically rare, the dissociation of water molecules has a great effect on organisms |
In pure water 554 million water molecules dissociated |
|
|
Concentrations of H+ OH- |
Are equal in water |
|
|
Acid |
Any substance that increases the H+ concentration of a solution |
|
|
Base |
Any substance that reduces the H+ concentration of a solution |
|
|
Examples of pH levels (pic) |
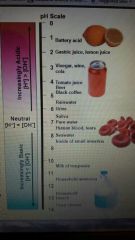
|
|
|
Buffers |
Substances that minimize changes in concentrations of H+ and OH- in a solution |
|
|
Fossil fuels |
CO2 is a main product of fossil fuel combustion. 25% of human generated CO2 is absorbed by the oceans. |
|
|
Ocean acidification |
Yhe proccess in which CO2 is dissolved in sea water forms carbon acid |
|
|
hydrophilic |
ionic and polar substances; they have an affinity for water due to electrical attractions and hydrogen bonding |
|
|
hydrophobic |
non-polar and nonionic substances; they will not easily mix with or dissolve in water |
|
|
molecular mass |
the sum of the mass of all atoms in the molecule |
|
|
mole (mol) |
is the amount of a substance that has a mass in grams numerically equivalent to its molecular mass. |
|
|
molarity |
refers to the number of moles of a solute dissolved in 1 liter of solution |
|
|
buffers |
regulate an organism's pH |
|
|
hydrogen bonding between polar water molecules |
creates a cohesive liquid with a high specific heat and high heat vaporization, both of which help to regulate environmental temperature. |
|
|
Ice |
floats and protects oceans and lakes from freezing |
|
|
The polarity of water |
makes it versatile solvent |
|
|
The H+ in a solution |
is expressed as pH and determines if a solution is acidic, neutral or basic |
|
|
A water molecule consists of |
two hydrogen atoms, each bonded to a more electronegative oxygen atom by a polar covalent bond |
|
|
the polar molecule |
has a V shape with partial positive charge on each hydrogen atom and a partial negative charge associated with the oxygen |
|
|
Creates a structural organization that leads to emergent properties of water? |
Hydrogen bonds |
|
|
hydrogen bonds are |
electrical attractions between the hydrogen atom of one water molecule and the oxygen atom of a nearby water molecule |
|
|
how many water molecules can hydrogen bond to one water molecule? |
four |
|
|
thermal energy is a measure of |
kinectic energy (the energy of motion) associated with the random movement of atoms and molecules. |
|
|
temperature measures |
the average kinetic energy of molecules in a body of matter
|
|
|
thermal energy reflects |
total kinetic energy in that matter, which relates to volume of body of matter. |
|
|
the thermal energy that transfers from warmer to cooler body of matter is defined as |
heat |
|
|
How is heat measured? |
a calorie calorie is the amount of heat it takes to raise 1 g of water 1 degree celsius. A kilocalorie is 1,00 calories, the amount of heat required or released to change the temperature of 1 kg of water by 1 degree Celsius. A joule equals 0.239 cal; one calorie is 4.184 j |
|
|
a calorie (cal) is? |
the amount of heat it takes to raise 1g of water1 degree celsius |
|
|
What is the amount of amount absorbed or lost when 1 g of a substance changes its temp by 1 degree celsius? |
Specific heat |
|
|
What is water's specific heat of 1 cal/g degrees celsius? |
is unusually high compared with other common substances |
|
|
why does water absorb or release a relatively large quantity of heat as its temperature changes? |
heat must be absorbed to break hydrogen bonds, before water molecules can move faster and the temperature can rise, and conversely, heat is released when hydrogen bonds form as water molecules slow down when the temperature of water drops. |

