![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
60 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
|
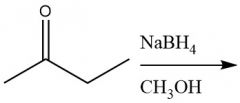
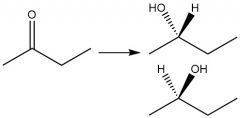
Borohydride Ketone Reduction (20.4): Can attack aldehydes and ketones |
|
|
|
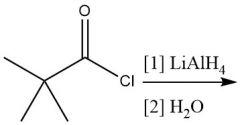
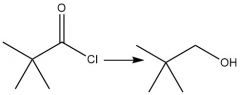
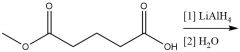
Nucleophilic Acyl Substitution (20.7): RCOCl → RCH₂OH R₂CO→R₂CHOH |
|
|
|
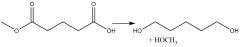
Organometallic Reduction (20.7) Alum. Hydride Carboxyl Reduction (20.7) |
|
|
|
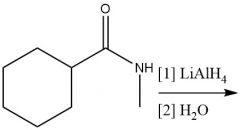
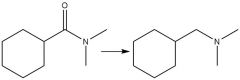
Alum. Hydride Amine Reduction (20.7) |
|
|
|
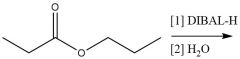
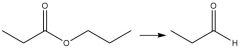
DIBAL-H Reduction: Use to reduce esters Must be run at -78°C |
|
|
|
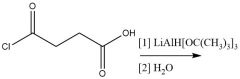
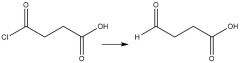
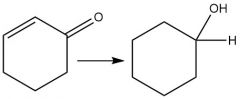
"Lithium tri-tert-butoxyaluminum hydride" Reduction |
|
|
|
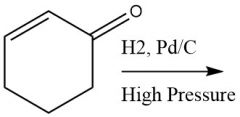
High-Pressure Catalytic Metal Reduction (20.4C) |
|
|
|
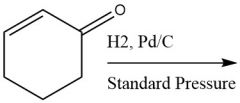
Standard-Pressure Catalytic Metal Reduction (20.4C) |
|
|
|
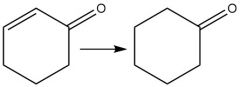
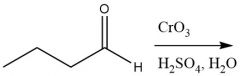
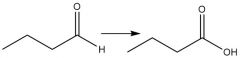
Cr⁶⁺ Oxidation of Aldehydes (20.8): Oxidizes alcohols similiarly |
|
|
|
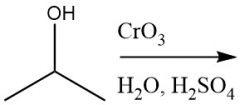
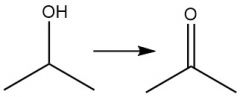
Cr⁶⁺ Oxidation of Alcohols (12.12)
|
|
|
|
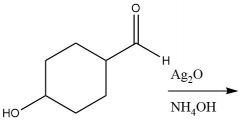
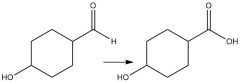
Tollens Reagent (20.8): Oxidation of aldehydes No mechanism Selectively oxidize aldehyde to carboxylic acid |
|
|
|
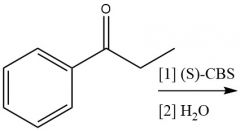
Enantioselective Carbonyl Reduction (20.6): |
|
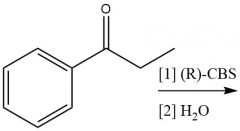
|
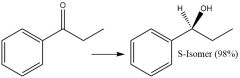
|
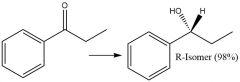
Enantioselective Carbonyl Reduction (20.6): |
|
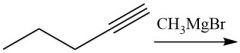
|
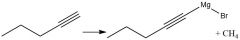
|
Deprotanation Grignard (20.9) |
|
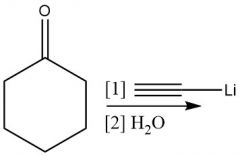
|
|
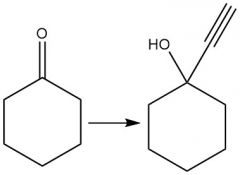
Organometallic new C-C bond (20.10) |
|
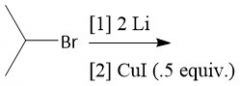
|
|
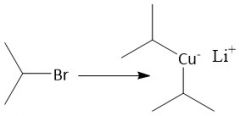
Organocuprate Synthesis (20.9) |
|
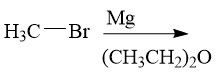
|
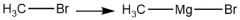
|
Gignard Reagent Synthesis (20.9) |
|
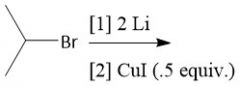
|
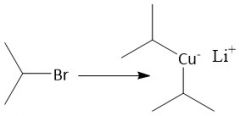
|
Organolithium Synthesis (20.9) |
|
|
|
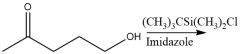
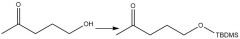
Protection (20.12): tert-butyldimethylsilyl |
|
|
|
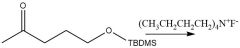
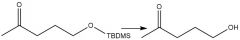
TBAF Deprotection (20.12): Tetra-n-butylammonium fluoride |
|
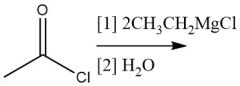
|
|
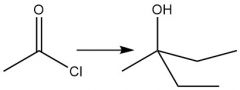
Organometallic Dialkylation (20.13) Acid Halide |
|
|
|
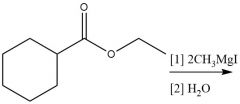
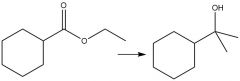
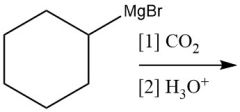
Grignard Nuceophilic Acyl Substitution (20.13): MUST use (2 equiv.) |
|
|
|
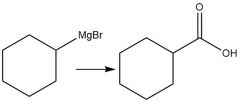
Carboxylation (20.14) |
|
|
|
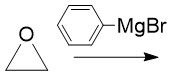
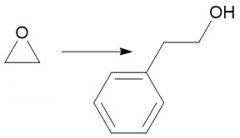
Expoxide Opening (20.14) |
|
|
|
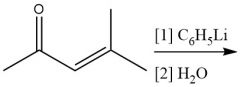
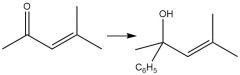
1,2-Addition Grignard (20.15) |
|
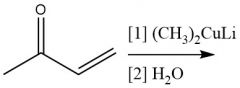
|
|
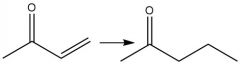
1,4-Addition Organocuprate (20.15): Uses Enolate path Organocuprates add 1,4 |
|
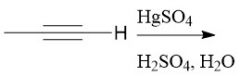
|
|
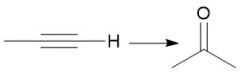
Alkyne Hydration (11.9) |
|
|
|
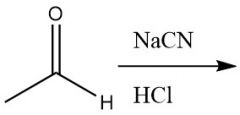
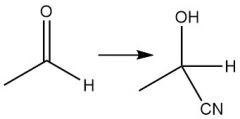
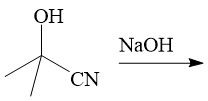
Cyanohydrin Formation (21.9) |
|
|
|
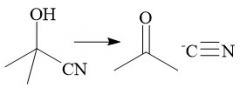
Cyanohydrin Revertion (21.9) |
|
|
|
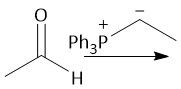
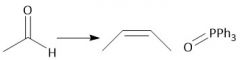
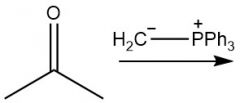
Wittig Formation (21.10): Z isomer (standard) |
|
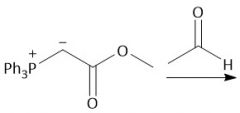
|
|
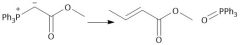
Wittig Formation (21.10): E Isomer (conjugated) CAREFUL: Make sure there are no unprotected alcohols or carboxy groups on your wittig! |
|
|
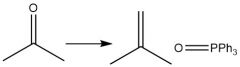
|
Wittig Reaction (21.10): [1] Ylide attacks carbonyl Oxaphosphetane Inter. Forming a 1° halide is easier than forming a 2° Adds Z unless the product is conjugated than it forms E |
|
|
|
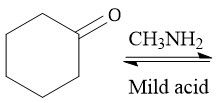
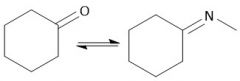
Imine Formation (21.11): Formed from 1° amine Carbinolamine interm. Iminium Ion interm. Also called a Schiff base Requires pH 4-5 Every step is in equilibrium |
|
|
|
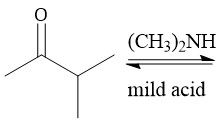
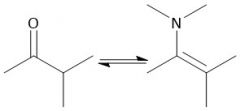
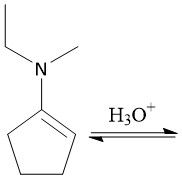
Formation of Enamines (21.12): Formed from 2° amine Forms E and Z isomers |
|
|
|
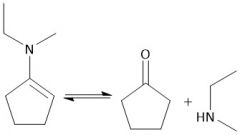
Enamine Hydrolysis (20.12) |
|
|
|
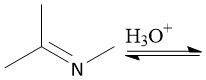
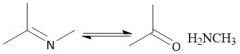
Imine Hydrolysis (20.12) |
|
|
|
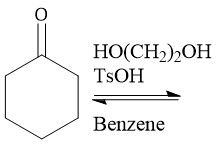
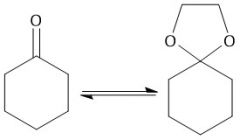
Dioxalane Protection (21.14): Carbonyl protection Hemiacetal formation Only occurs with ketones and aldehydes |
|
|
|
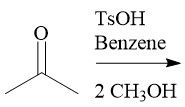
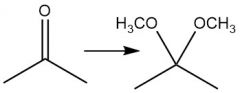
Acetal Formation (21.14): Also with aldehydes Reversible |
|
|
|
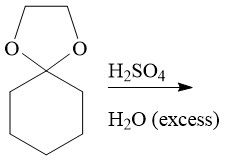
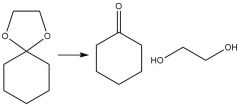
Hydrolysis (21.14): Le Chatelier's Principle Requires a strong acid Carbonyl deprotection Reversible |
|
|
|
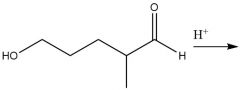
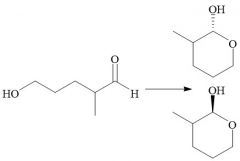
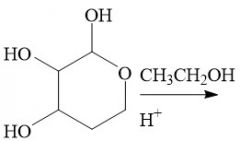
Intramolecular Hemiacetal Formation (21.16): Faster than intermolecular rxns. Cyclic 5 or 6 carbon rings Axial and equatorial isomers Requires aldehydes Reversible |
|
|
|
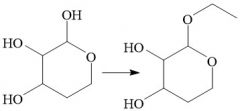
Hemiacetal to Acetals (21.16) Reversible |
|
|
|
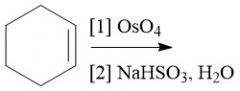
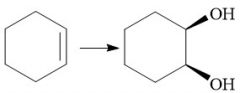
Oxidation |
|
|
|
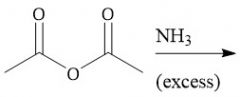
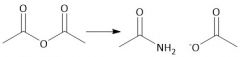
Anhydride Amidification (22.9) |
|
|
|
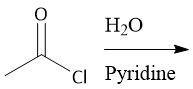
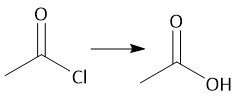
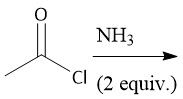
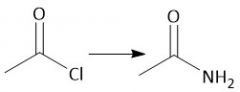
Acid Chloride Hydrolysis (22.8) |
|
|
|
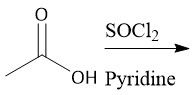
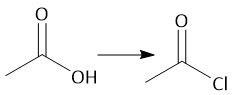
Acid Chloride Amidification (22.8): Also works with alcohols |
|
|
|
Thionyl Chloride as Nu (22.10): Pyridine deprotonates NR with anhydrides |
|
|
|
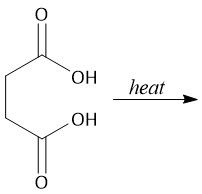
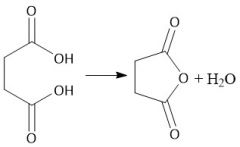
Intramolecular Anhydride Formation (22.10B) |
|
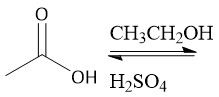
|
|
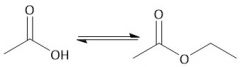
Fischer Esterification (22.10C): Will not work in the presence of a base (forms carboxylate anion) Use strong acid |
|
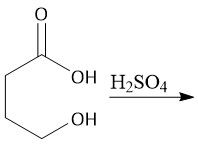
|
|
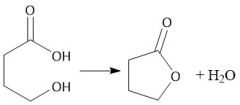
Intramolecular Esterification (22.10C) |
|
|
|
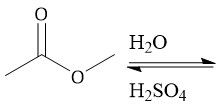
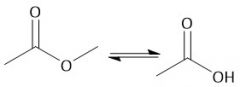
Acidic Ester Hydrolysis (22.11) |
|
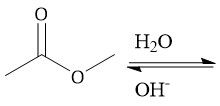
|
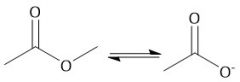
|
Saponification (22.11): Basic ester hydrolysis Resonance-stabilized carboxylate anion Protonate with Strong HA Not reversible |
|
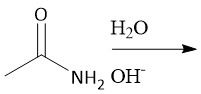
|
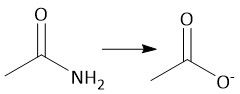
|
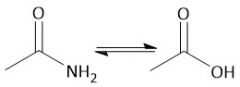
Base-Catalyzed Amide Hydrolysis (22.13): Not reversible |
|
|
|
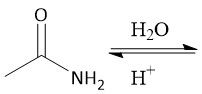
Acid-Catalyzed Amide Hydrolysis (22.13) |
|
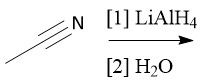
|
|
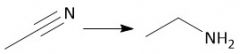
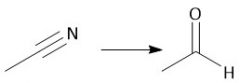
Cyano Reduction (22.18) |
|
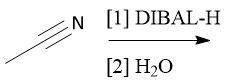
|
|
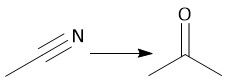
Hindered Cyano Reduction (22.18) |
|
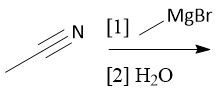
|
|
Cyano Gringard Rxn. (22.18) Only adds 1 methyl |
|
|
|
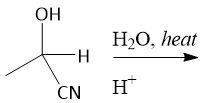
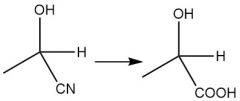
Acidic Cyanohydrin Hydrolysis (21.9) |
|
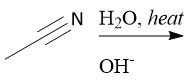
|
|
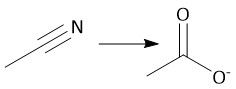
Basic Cyano Hydrolysis (21.9): [2] Imidic Acid [3-4] Tautomerization to Amide [2] Amide hydrolysis |
|
|
IR Absorption: Acid Chloride Anhydride Ester Amide |
1800 cm⁻¹ 1820-1760 cm⁻¹ 1735-1745 cm⁻¹ 1630-1680 cm⁻¹ |
|
|
|
H NMR Absorptions: Amide N-H Carbonyl Alpha C-H |
7.8-8.5 ppm 2-2.5 ppm |
|

