![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
28 Cards in this Set
- Front
- Back
|
In an exergonic process, the system |
Loses energy |
|
|
The concept of entropy |
Is employed as an indicator of disorder in a system |
|
|
Which of the following states of matter generally has the lowest entropy? |
A crystalline solid |
|
|
Which of the conditions given is necessary for a chemical reaction to occur |
The molecules of the reaction time of month bump into each other |
|
|
The reaction conditions for a specific reaction, BC D + ha ---》 ABCD h Karma are correct, but the reaction does not occur what could be the reason? |
The orientation of molecules with respect to each other is not correct for the reaction to occur |
|
|
Which of the following is most closely related to the term reaction rate |
The speed of a reaction |
|
|
Catalytic action is |
Directly opposed by inhibitors |
|
|
Sulfur reacts with oxygen to produce sulfur dioxide, but only in the high activation energy is supplied. Which condition will lower the activation energy the most? |
Using a catalyst |
|
|
The energy required to start some spontaneous process is called |
Activation energy |
|
|
In a system of equilibrium, it is true that the rate of the forward reaction |
Is equal to the rate of the reverse reaction |
|
|
Which statement applies to equilibria in general? |
The larger the value of K, the greater the amount of matter on the right |
|

|
Shift right |
|

Refer to equilibrium. Remove H2 from the mixture |
Shift right |
|

Refer to the equilibrium. Add a catalyst to the mixture. |
No effect |
|

When the equilibrium expression is written for the reaction. What is the exponent of the concentration of fluorine, F2? |
3 |
|
|
Which of the following statement about A K of 3.5 calculateed for a specific chemical reactions at equilibrium is incorrect? |
A K of 3.5 indicates that a catalyst will push the reaction to the right |
|
|
Which statement is correct application of La chatelier's principle? |
Additions of a substance to the left side of an equilibrium shifted to the right |
|
|
A process that gains or accept energy as it takes place is a |
Endergonic process |
|
|
I has a ____ entropy then that of liquid water |
Lower |
|
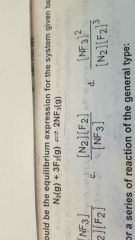
Which of the following would be the equilibrium expression for a system given below? |
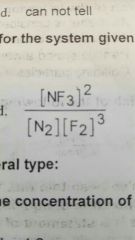
|
|

St.clement consist for a series of reactions of the general type |

|
|

|
Decreasing the temperature |
|

|
1 |
|

|
C |
|
|
Which of the following is not true regarding a chemical reaction? |
All particles contained potential energy, and measured by a constant motion |
|

|

|
|
|
You been told that when a change is made in any factor of an established equilibrium the position of equilibrium will shift in a direction that will minimize or oppose the change. This is a statement of which of the following |
Le chatelier's principle |
|
|
You purchase a glow stick during an evening parade. After a 10, your roommate tells you that if you place it in the freezer, it will go for a longer time. Assuming she knows what she's talking about, why would this be true |
Reaction rates can be expected to decrease as temperature is reduced |

