![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
78 Cards in this Set
- Front
- Back
|
Name of the number at the bottom of the chemical symbol |
The atomic number |
|
|
Name of the number at the top of the chemical symbol |
The mass number |
|
|
How do you find the number of electrons with the atomic number |
It's the same |
|
|
How is a compound formed |
When atoms of two or more elements are chemically combined together |
|
|
Definition of an isotope |
Different atomic forms of the same element which have the same number of protons but a different number of neutrons |
|
|
Give an example of a pair of isotopes |
Carbon-12 and Carbon-14 |
|
|
Which type of bonding is the transferrance of electrons |
Ionic |
|
|
Why do elements such as sodium, potassium and calcium like to form ionic bonds |
They have 1 electron in their outer shell |
|
|
Which two groups contain elements which are most likely to form ionic bonds by gaining electrons |
Groups 6 & 7 |
|
|
What kind of structure do ionic compounds have |
Regular lattice |
|
|
Why do ions 'stay together' in a lattice structure |
Because of the strong electrostatic forces of attraction between oppositely charged ions in all directions |
|
|
Name an example of a compound in a lattice structure |
Sodium chloride |
|
|
Name the properties of ionic compounds |
- High melting points |
|
|
Why do ionic compounds have high melting and boiling points |
Because of the strong attraction between the ions. It takes a large amount of energy to overcome this. |
|
|
Which groups are most likely to form ions |
Groups 1 & 2 and Groups 6 & 7 |
|
|
Ions have the structure of what |
A noble gas |
|
|
Which type of bond is the sharing of electrons |
Covalent |
|
|
Draw a diagram for the covalent bond of hydrogen |

|
|
|
Draw a diagram for the covalent bond of chlorine |
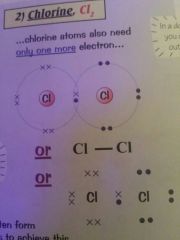
|
|
|
Draw a diagram for the covalent bond of methane |

|
|
|
Draw a diagram for the covalent bond of hydrogen chloride |

|
|
|
Draw a diagram for the covalent bond of ammonia |

|
|
|
Draw a diagram for the covalent bond of water |

|
|
|
Draw a diagram for the covalent bond of oxygen |

|
|
|
Name the two types of substances with covalent bonds |
- Simple molecules |
|
|
Why are the melting and boiling points of simple molecular substances low |
Because of weak intermolecular forces. The molecules are easily parted from each other. |
|
|
What happens when simple molecular substances melt or boil |
Intermolecular forces are broken |
|
|
What state are most molecular substances in at room temperature |
Gases or liquids |
|
|
Why don't molecular substances conduct electricity |
There are no ions so there's no electrical charge |
|
|
What is a synonym for giant covalent structures |
Macromolecules |
|
|
What is a lattice |
A giant ionic structure |
|
|
Why are macromolecules different from lattices |
Macromolecules have no charged ions |
|
|
How are atoms bonded together in macromolecules |
Strong covalent bonds |
|
|
Features of macromolecules? |
- high melting & boiling points |
|
|
3 examples of macromolecules? |
- Diamond |
|
|
How many covalent bonds are formed by each carbon atom in diamond |
4 |
|
|
What is diamond used for |
Drill tips |
|
|
What is sand made of |
Silicon dioxide |
|
|
In graphite how many covalent bonds are formed by each carbon atom |
3 |
|
|
What do the three covalent bonds formed by each carbon atom in graphite create |
Layers which are free to slide over each other |
|
|
What features are created in graphite by the layers formed |
Makes it soft and slippery |
|
|
How does a pencil work |
The layers are held together so loosely in graphite that they can be rubbed off on to paper |
|
|
Why can layers be rubbed off on to paper in graphite |
Because there are weak intermolecular forces between layers |
|
|
Why is graphite a good conductor of heat and electricity |
Each carbon atom has one delocalised electron which conducts it |
|
|
What are the properties of metals due to |
The sea of free/delocalised electrons |
|
|
Where do the delocalised electrons come from |
The outer shell of every metal atom in the structure |
|
|
Why are metals good conductors of heat and electricity |
Because the electrons are free to move throughout the structure |
|
|
Why are atoms in metals held together in a regular structure |
There are strong forces of electrostatic attraction between the positive metal ions and the negative electrons |
|
|
What does the sea of free electrons allow to happen in metals |
The layers of atoms can slide over each other allowing metals to be bent and shaped |
|
|
Which are harder; pure metals or alloys |
Alloys |
|
|
What is an alloy |
A mixture of two or more metals |
|
|
Why are alloys harder than pure metals |
Different elements have different sized atoms. The new metal atoms will distort the layers of metal atoms making it more difficult for them to slide over each other. |
|
|
Name the 4 types of structures |
- Giant ionic |
|
|
What do smart materials do |
Behave differently depending on the conditions |
|
|
Give an example of a smart material |
Nitinol, a shape memory alloy |
|
|
State the properties of nitinol |
- When cool it's bendable and you are able to twist it like rubber |
|
|
Give two examples of uses for nitinol |
- Glasses frames, if accidentally bent they can be put in a bowl of hot water and they'll jump back into shape. |
|
|
What are nanoparticles |
Tiny particles, 1-100 nanometres across |
|
|
How many atoms do nanoparticles contain |
Roughly a few hundred |
|
|
Nanoparticles include what |
Fullerenes |
|
|
What are fullerenes |
Molecules of carbon, shaped like hollow balls or closed tubes. |
|
|
What is a nanotube |
Fullerenes joined together. |
|
|
Which type of bond in nanotubes make them strong |
Covalent |
|
|
Give an example of an object in which nanotubes are used |
Used to reinforce graphite in tennis rackets |
|
|
Using nanoparticles is known as |
Nanoscience |
|
|
List possible uses for nanoparticles |
- Catalysts due to large surface area to volume ratio |
|
|
Explain the idea behind nanomedicine |
Tiny fullerenes can be absorbed more easily by the body than most particles. Drugs can then be delivered right into the cells where they're needed. |
|
|
Why can nanotubes be used in electric circuits |
They conduct electricity |
|
|
Why can fullerenes be used to aid artificial joints and gears |
Lubricant coatings are being developed using fullerenes. They reduce friction a bit like ball bearings so can be used in artificial joints etc. |
|
|
What determines the properties of plastics |
Forces between molecules |
|
|
What holds the atoms together in long chains in plastics |
Strong covalent bonds |
|
|
Describe polymers with weak forces |
Individual tangled chains, held together by weak intermolecular forces which are free to slide over each other. |
|
|
Describe polymers with strong forces |
Strong intermolecular forces between polymer chains called crosslinks, that hold the chains together. |
|
|
Why can thermosoftening plastics be melted |
They don't have cross-linking between chains. The forces between them are really easy to overcome. |
|
|
Why can't thermosetting plastics be melted |
They have crosslinks which hold the chains together in a solid structure. |
|
|
Give examples of two types of polythene made using different conditions |
- Low density polythene |
|
|
How is LDPE made & what are its properties |
By heating ethene to about 200 degrees C under high pressure. |
|
|
How is HDPE made & what are its properties |
Made at a lower temperature than LDPE and pressure (with a catalyst). |

