![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
45 Cards in this Set
- Front
- Back
|
Which of the following is NOT a mineral? |
All of these |
|
|
Which of the following is a mineral? |
Ice |
|
|
Which of the following is true about rocks and minerals? |
All of these |
|
|
Which of the following is NOT a required criterion for a substance to be classified as a mineral? |
human made |
|
|
Rocks are composed of: |
minerals |
|

What type of rock is shown in this photograph? |
Crystalline rock |
|

What type of rock is shown in this photograph? |
Clastic |
|
|
What are the two main ways that minerals are put together in rocks? |
Crystalline and Clastic |
|
|
Important characteristics to observe in rocks include: |
all of the above |
|
|
Which of the following is NOT a characteristic of rocks with clastic textures? |
composed of interlocking minerals |
|

What mineral property is illustrated by this photograph? |
color |
|
|
Which mineral property is the least reliable for identifying a mineral? |
Color |
|
|
Which of the following is true about tests we can perform on a mineral? |
none of these |
|
|
A mineral property that can be observed without using a test is: |
Cleavage |
|

What mineral is shown in this photograph? |
Magnetite |
|

What mineral test is shown in this photograph? |
magnetic |
|

What mineral is shown in this photograph? |
calcite |
|

What mineral test is shown in this photograph? |
acid |
|

What mineral test is shown in this photograph? |
streak |
|
|
A cube of some rock weighs 6 grams, and a same-sized cube of water weighs 2 grams. What is the specific gravity of this rock? |
3 |
|
|
Identify the list that consists of ten common minerals ranked in order of increasing hardness, from 1 to 10. |
Moshs Hardness scale |
|
|
The ratio of density of a mineral to that of freshwater is called the: |
specific gravity |
|
|
Which of the following are tests we can quickly and easily perform (even in the field) to help us identify a mineral? |
all of these |
|
|
Which of the following is NOT a main group of chemical elements on the Periodic Table? |
sulfides |
|
|
Which of the following is NOT a major class of rock-forming minerals? |
argonites |
|
|
The most important class of rock-forming minerals on Earth is: |
Silicates |
|
|
Which of the following is NOT a main type of silicate structure? |
triple chains |
|

What type of silicate minerals is shown from this arrangement of silicon tetrahedron? |
Single chains |
|
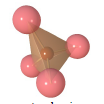
What type of atom is represented by the small brown sphere in the center of this silica tetrahedron? |
silicon |
|
|
The dominant shape in which silicon and oxygen atoms combine in silicate minerals is: |
none of these |
|
|
How do silica tetrahedra bond in silicate minerals? |
All of these |
|
|
Which of the following is generally a light-colored silicate mineral? |
Quartz |
|
|
Which of the following minerals is typically green and has no cleavage? |
Olivine |
|
|
Which of the following is NOT composed of calcium carbonate? |
Pyrite |
|
|
Which of the following is a silicate mineral? |
Garnet |
|
|
Minerals that do not contain silicon are classified as: |
Non-Silicate |
|
|
The common nonsilicate mineral groups include: |
All of these |
|

Which layer in the earth has a composition similar to the granite shown in this photograph? |
Continental crust |
|

Which layer in the earth has a composition similar to the green olivine in the center of this rock? |
mantle |
|
|
The two elements most abundant in the universe are: |
Hydrogen and Helium |
|
|
The two elements most abundant in the earth's crust are: |
Oxygen and Silicon |
|
|
What three kinds of particles are the main building blocks of an atom? |
Proton, Neutron and Electron |
|
|
The measure of an element's ability to attract electrons is its: |
Electronegativity |
|
|
What characteristic of water help it cause a mineral, such as halite, to dissolve? |
The Polar nature of water molecule |
|
|
What is a diagnostic characteristic of a mineral used to make a cement slab on your house? |
it reacts with dilute hydrochloric acid |

