![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
23 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
What is the Kinetic Theory of matter? |
This speaks to tiny particles that are in constant motion and these particles are atoms, molecules and ions. |
Constant motion |
|
|
What are the two things that determine the amount of kinetic energy held in matter? |
State and Temperature |
|
|
|
What are the 3 phenomenas that provide evidence of kinetic Theory? |
Diffusion Osmosis Brownian Motion |
|
|
|
What is diffusion? |
This the spontaneous movement of particles from a region of high to low concentration until evenly distributed. |
Evenly distributed |
|
|
What is osmosis? |
The movement of water particles from an area of high to low concentration through a semi-permeable (selective) membrane. |
Semi-permeable membrane |
|
|
Brownian Motion |
This the visible movement of small pieces of solid that are big enough to be seen under a microscope. |
Visible movement |
|
|
Is diffusion in gases faster than liquids? Why |
Diffusion in gases is faster than in liquids. This is because the particles in gases move faster than in liquids. There is little or no movement in solids. |
|
|
|
Explain the diffusion experiment |
The hydrogen chloride and ammonia particles are in constant motion. They bump and bounce of each other along with the air. A white ring is formed where the gases meet.
The white ring is ammonium chloride. The white ring is nearer to the hydrogen chloride because it is a heavier molecule than ammonia molecule. Heavier molecule move more slowly than lighter ones. |
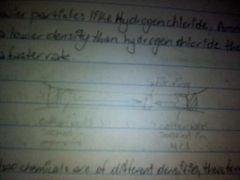
|
|
|
Speed of diffusion |
The speed of diffusion is faster in gases followed by liquids and very slowly in solids. The speed of diffusion increases with temperature. At higher temperatures, the particles have higher kinetic energy and hence move faster and diffuse at a faster rate. Heavier particles diffuse slower than lighter particles. The speed of diffusion also depends on the relative molecular mass of the particles. |
|
|
|
Distance/ Travel time |
The speed of diffusion in a gas depends on its density. |
|
|
|
Osmosis in paw paw (distilled water) |
The water outside the cells is more concentrated than inside the cells.
Water molecules passes into the cells by osmosis, resulting in the strips becoming longer and more rigid. |
|
|
|
Osmosis in paw paw (sugar solution) |
The water outside the cell is less concentrated than water inside the cell. This is because there is sugar and water in the solution. Water molecules pass out of the cells by osmosis resulting in the strip becoming shorter and flaccid. |
|
|
|
What is the nature of matter? |
The nature of matter is that it moves from an area of high to low concentration. As well as, particles in matter are always in constant motion due to the kinetic energy that they posses. |
|
|
|
Describe the volume, shape and compressibility of solid. |
Volume - fixed Shape - fixed Compressibility - very difficult to compress |
|
|
|
Describe the volume, shape and compressibility of liquid. |
Volume - fixed Shape - take shape of the bottom of the container. Compressibility - slight |
|
|
|
Describe the volume, shape and compressibility of gas |
Volume - variable, expands to fill the whole container. Shape - takes shape of the whole container. Compressibility - easily compressed. |
|
|
|
Describe the three states of matter. |
Solid - particles are very closely together in every orderly pattern. Liquid - particles are further apart. The arrangement is less orderly. Gas - particles are very far apart and are haphazardly arranged. |
|
|
|
Describe the forces of attraction of each state of matter |
Solid - the forces of attraction between particles are strong so that the movement is limited to vibration around a fixed position. Liquid - the forces of attraction are weaker so the particles are able to slip slowly over each other. Gas - the forces of attraction between particles are so small that particles are able to move freely in all directions at high speeds. |
|
|
|
Particle Theory |
Gas particles of a substance have more kinetic energy than liquids particles, while liquid particles particles have more kinetic energy than solid particles. Matter can be converted from one state to another by changing the amount of energy of the particles. The more heat energy, the faster the particles will move. If heat energy is removed from a matter the particles will slow down and come closer together. |
|
|
|
Changes of state |
Melting - the change from solid to liquid. The temperature. The temperature at which a solid melts is called its melting point. Freezing - the change from a liquid to a solid. The temperature at which a liquid freezes is called its freezing point. For pure substances, the freezing point is the same as the melting point. Boiling - is the change from a liquid to a gas or vapour. The temperature at which a liquid boils is called its boiling point. A pure substance has a fixed boiling point. Evaporation - the change from liquid into gas without boiling. |
|
|
|
Changes of state 2 |
The change from gas to liquid. It is the reverse of boiling. Heat energy is released during condensation as the gas particles slow down and move closer together to become liquid. Sublimation - the change from a solid into a gas without melting. Deposition or desublimation - is the reverse of sublimation. |
|
|
|
What is the Heating curve |
A heating curve is drawn when a temperature of a solid is measured at intervals as it is heated and changes state to the liquid and then to a gas, and the temperature is then plotted against time. |
|
|
|
What is the cooling curve ? |
Cooling curve is drawn when the temperature of a gas is measured at intervals as it is cooled and changes state to a liquid and then to a solid and the temperature is then plotted against time. |
|

