![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
98 Cards in this Set
- Front
- Back
|
What is the goal of antiepilectic drugs? (AEDs)
|
Goal: to suppress the formation or spread of abnormal electrical discharges in the brain
-Inhibition of Na+ or Ca2+ influx responsible for neuronal depolarization -Increase inhibitory neurotransmission by GABA (opening of Cl- channels) -Inhibit excitatory glutamate neurotransmission |
|
|
AEDs targeting the excitatory, glutamatergic synapse: 1, voltage-gated (VG) Na+ channels
|
phenytoin, carbamazepine, lamotrigine, and lacosamide
|
|
|
AEDs targeting the excitatory, glutamatergic synapse: 2, VG-Ca2+ channels
|
ethosuximide, lamotrigine, gabapentin, and pregabalin
|
|
|
AEDs targeting the excitatory, glutamatergic synapse: 3, K+ channels
|
retigabine
|
|
|
AEDs targeting the excitatory, glutamatergic synapse: synaptic vesicle proteins, 4, SV2A
|
levetiracetam
|
|
|
AEDs targeting the excitatory, glutamatergic synapse: 5, CRMP-2, collapsin-response mediator protein-2
|
lacosamide
|
|
|
AEDs targeting the excitatory, glutamatergic synapse: 6, AMPA receptors blocked by:
|
phenobarbital, topiramate and lamotrigine
|
|
|
AEDs targeting the excitatory, glutamatergic synapse:
7, NMDA receptors blocked by: |
felbamate
|
|
|
AEDs targeting the inhibitory GABAergic synapse:
1, GABA transporters (especially GAT-1) |
tiagabine
|
|
|
AEDs targeting the inhibitory GABAergic synapse:
2, GABA-transaminase (GABA-T) |
vigabatrin
|
|
|
AEDs targeting the inhibitory GABAergic synapse:
3, GABAA receptors and 4, potentially GABAB receptors |
benzodiazepines
|
|
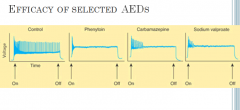
Flip the card to learn some stuff
|
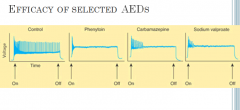
In the absence of drug, a series of high-frequency repetitive action potentials filled the entire duration of the current pulse. Phenytoin, carbamazepine, and sodium valproate all markedly reduced the number of action potentials elicited by the current pulses
|
|
|
Which two AEDs are NOT bound to plasma proteins?
|
General Pharmacokinetics
-Similar pharmacokinetics though structurally and chemically different -Most are orally active; 80-100% of [drug] into circulation --Most not highly bound to plasma proteins ---**EXCEPTIONS: Phenytoin and Valproic Acid -Predominantly distributed into total body water -Plasma clearance is slow --Most considered to be medium- to long- acting |
|
|
More general PK... Is this class of drugs black boxed?
|
The ENTIRE class is black boxed
-Cleared through hepatic mechanisms --Metabolites (also active) cleared through liver -Most older AEDs are inducers of the cytochrome P450 enzymes --Many are substrates --Few are inhibitors Many drug interactions!!! |
|
|
Partial Seizures & Generalized Tonic-Clonic Seizures
|
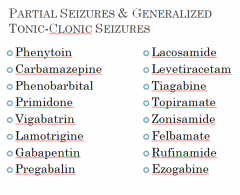
|
|
|
Generalized tonic-clonic seizures
|
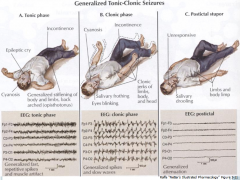
|
|
|
Phenytoin
|
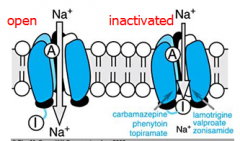
Mechanism of Action:
-Alters Na+, K+, Ca2+ conductance --Prolongs the INACTIVE state of the Na+ channel --Inhibits the generation of repetitive action potentials |
|
|
more Phenytoin
|
-Decreases glutamate release
-Increases GABA release **Active against: -Simple Partial -Complex Partial -Partial with secondarily generalized tonic-clonic |
|
|
Phenytoin PK
|
Given orally (phenytoin sodium)
2 forms -Extended-release --Dissolves slowly; once daily dosing -Rapid-release Absorption from GI tract is ~100% -Time to peak is variable (3-12 hrs) |
|
|
What is the more soluble form of phenytoin that can be used parenterally?
|
Fosphenytoin (parenteral)
-More potent than phenytoin -Rapidly converted to phenytoin |
|
|
Is phenytoin protein bound?
|
Highly bound by protein
Other highly bound drugs may displace bound phenytoin → increase in free drug Accumulates in brain, liver, muscle and fat |
|
|
What cytochrome P450 metabolizes phenytoin?
|
Metabolized to inactive compounds by *CYP2C9*/2C19
-Induces hepatic microsomal enzymes -CYP2C and 3A --2C9/3A4 Elimination is dose-dependent -Low blood levels – metabolism follows first order kinetics – no big deal -Increased blood levels – liver capacity to metabolize drug maxes out (saturation kinetics; zero order) |
|
|
What are the zero order drugs?
|
Phenytoin, EtOH & aspirin– the zero order drugs (PEA)
|
|
|
dose dependent kinetics
|

|
|
|
What competes with protein binding, increasing phenytoin plasma levels?
|
valproate
|
|
|
Phenytoin enztme interactions prevent degradation of what drug? (non-AED)
|
warfarin; can lead to bleeding disorders
|
|
|
What does phenytoin do to OCP metabolism?
|
enhances it
|
|
|
what drug enahnces phenytoins metabolism and vice-versa?
|
carbamazepine
|
|
|
phenytoin has an affinity for thyroid-binding globulin...
|
May confound thyroid function tests
Should measure TSH levels instead |
|
|
phenytoin toxicity
|
"DANG":
Diplopia, ataxia, nystagmus, gingival hyperplasia also: hirsutism, coarsening facial features, mild peripheral neuropathy, osteomalacia... **HTN and arrhythmias with rapid infusion** (black box) |
|
|
Carbamazepine
|
Mechanism of action:
Similar to Phenytoin -Blocks Na+ channels and inhibits high-frequency repetitive firing of neurons -Acts pre-synaptically to reduce synaptic transmission Clinical Uses Seizures -Simple Partial -Complex Partial -Partial with secondarily generalized tonic-clonic -Tonic-clonic Bipolar disorder Trigeminal neuralgia |
|
|
Can carbamazepine be given parenterally?
|
**ORAL ONLY**
Effective in children and adults Peak levels are achieved 6-8 hrs after administration Large doses are given after meals to slow absorption Half-life ~ 36 hours |
|
|
What cytochrome P450 does carbamazepine induce?
|
autoinduces CYP3A4, which increases its own metablism.
|
|
|
what is carbamazepine's active metabolite with anticonvulsant activity?
|
Carbamazepine-10,11-epoxide
|
|
|
Carbamazepine interactions
|
Metabolism may be decreased by:
-Valproic acid Metabolism may be increased by: -Phenytoin and phenobarbital Increased metabolism other drugs: EX: primidone, phenytoin, ethosuximide, valproic acid and clonazepam |
|
|
How does the autoinduction (CYP3A4) and 10,11-epoxide metabolite impact dosing of carbamazepine?
|
Because of this distinct elimination process, it is difficult to rely on the dose of carbamazepine for predicting the serum concentration (thus therapeutic affect) of either the parent drug or the active metabolite. It is recommended that therapeutic levels of the carbamazepine and metabolite (10,11-epoxide) be monitored in both free and protein-bound states (lexi-comp)
|
|
|
Carbamazepne Toxicity
|
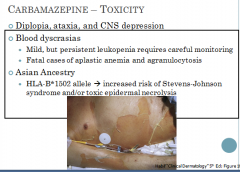
|
|
|
What patients are most affected by blood dyscrasias?
|
Most of the blood dyscrasias occur in the elderly taking the drug for trigeminal neuralgia and most occur within the first 4 months of treatment
|
|
|
What's the concern with generic carbamazepine?
|
This genetic variant has been associated with a significantly increased risk of developing Stevens-Johnson syndrome and/or toxic epidermal necrolysis. Patients with a positive result should not be started on carbamazepine.
|
|
|
How does phenobarbital work?
|
Unknown mechanism of action
**Enhancement of inhibitory neurotransmission -Binds to the GABAA receptor prolonging the opening of the Cl- channel Diminished excitatory neurotransmission -Effect is on presynaptic glumate release May inhibit the spread of abnormal firing from the seizure foci |
|
|
What are the phenobarbital clinical indications?
|
Partial with secondarily generalized tonic-clonic
Tonic-clonic Uncontrollable seizures of all types |
|
|
When is phenobarb a DOC?
|
infants
|
|
|
For what kind of seizures is phenobarb not effective?
|
absence, atonic attacks and infantile spasms
|
|
|
Primidone is a parent drug... what are the 2 active metabolites?
|
phenobarbital and phenylethylmalonamide (PEMA)
|
|
|
Primidone MOA?
|
Blocks voltage-sensitive Na+ channel (like phenytoin)
Potentiates GABA (formation of phenobarbital) |
|
|
What are the clinical indications for primidone?
|
Seizures in infants and elderly
Primidone is slowly metabolized Simple partial Partial with secondarily generalized tonic-clonic Tonic-clonic |
|
|
How is primidone metabolized?
|
Metabolized by oxidation to phenobarbital
Accumulates slowly Metabolized by scission to form PEMA Both phenobarb and PEMA undergo subsequent conjugation and excretion |
|
|
what happens if you start a patient on primidone at regular dosing?
|
Must start doses small and increase over weeks to avoid GI upset and sedation
Dose adjustments must consider steady state of parent drug (rapid) and metabolites (days to weeks) |
|
|
ViGABAtrin: how does it word?
|
GABA
-Irreversible inhibitor of GABA aminotransferase (GABA-T) --Enzyme responsible for degrading GABA -Inhibition of the vesicular GABA transporter -Decreased brain glutamine synthetase activity |
|
|
what are the clincal indications of Vigabatrin?
|
Infantile Spasms
Refractory Complex Partial |
|
|
What is the major toxicity (black box) for Vigabatrin?
|
Vision Loss – permanent!
|
|
|
Why can only specific physicians prescribe vigabatrin?
|
SHARE program
|
|
|
Lamotrigine MOA
|
Suppresses rapid firing of neurons
Produces a voltage- and use-dependent inactivation of Na+ channels Inhibits voltage-gated calcium channel (VGCC) May also decrease the release of glutamate |
|
|
Lamotrigine Clinical Use
|
Can be **adjunctive or monotherapy (for partial)
Absence and myoclonic seizures in children |
|
|
What is the notable AE of Lamotrigine?
|
skin rash
Black Box: Life-threatening dermatitis in 1-2% of pediatric patients |
|
|
Gabapentin and Pregabalin: Which GABA receptor do they activate?
|
Neither!
Structurally similar to GABA DO NOT DIRECTLY activate the GABA receptor Mechanism: **Both block voltage-gated Ca2+ channels Modify the release of GABA Decrease release of glutamate at synapse |
|
|
Gabapentin and Pregabalin – Clinical Use
|
Both typically given as adjunct therapy
-Gabapentin – partial and generalized tonic-clonic -Pregabalin – partial Both can also treat post-herpetic neuralgia |
|
|
Anything specail about Gabapentin AEs?
|
Not really.
somnolence, dizziness, ataxia, headache and tremor |
|
|
Why does the DEA care about Pregabalin?
|
Pregabalin: Schedule V (DEA) for euphoria
|
|
|
Lacosamide: MOA: Enhanced, slow inactivation of the____ channel.
|
Enhanced slow inactivation of the Na+ channel
Blocks the actions of neurotrophic factors on axonal and dendritic growth |
|
|
What patient population does Lacosamide suit?
|
Approved for adjunctive therapy in patients 16+ y.o.
Clinical Indication: Partial Has no effect on CYP450s; drug interactions are negligible |
|
|
Levetiracetam MOA and Clinical Indications
|
Mechanism of action:
-Binds selectively to synaptic vesicle protein SV2A --Protein may modify the synaptic release of glutamate and GABA Clinical Indications: -Partial -Primary generalized tonic-clonic *Myoclonic seizures in juvenile myoclonic epilepsy |
|
|
What must be avoided when taking Levetiracetam?
|
Adverse Effects: somnolence, asthenia, dizziness and CNS depression (avoid EtOH
|
|
|
Tiagabine (tiaGABINe): MOA, Clinical Use and major AE
|
Mechanism of action:
-Inhibitor of GABA uptake in both neurons and glia --Prolonging activity and increasing tonic inhibition Clinical Uses: -Adjunctive therapy of partial seizures Adverse Effects: -CNS depression -Suicidal Ideation -Severe skin reactions --Stevens-Johnson Syndrome or Toxic Epidermal Necrolysis |
|
|
Topiramate: Blocks repetitive neuronal firing through blockade of ____ channels.
|
Mechanism of Action:
-Blocks repetitive neuronal firing through blockade of Na+ channels -Potentiates the inhibitory effects of GABA -May interfere with excitation of glutamate neurons -Weak carbonic anhydrase inhibitor Can be used monotherapy |
|
|
Other clinical indications for ________:
Lennox-Gastaut syndrome West’s syndrome Absence seizures Migraine headaches |
Topiramate
Other clinical indications: Lennox-Gastaut syndrome West’s syndrome Absence seizures Migraine headaches |
|
|
What is a nasty AE of Topiramate?
|
urolithiasis... some others:
Occur initially while establishing dose Somnolence, fatigue, dizziness, cognitive slowing, paresthesias, nervousness and confusion Acute myopia and glaucoma may require prompt drug withdrawal Metabolic acidosis |
|
|
Zonisamide: MOA, Clinical Indications
|
Mechanism of action:
-Inhibition of the Na+ and Ca2+ channels Clinical Uses: -Partial -Generalized tonic-clonic -Infantile spasms |
|
|
What two AEDs are weak carbonic anhydrase inhibitors?
|
topiramate and zonisamide
|
|
|
Felbemate: MOA, Clinical Indications
|
Mechanism of Action:
-Use-dependent block of NMDA receptor -Potentiates GABAA receptor response Clinical Indications – NEVER FIRST LINE -Refractory partial seizures -Lennox-Gastaut syndrome |
|
|
What are the two black box warnings for Felbemate?
|
Aplastic anemia
Severe hepatitis |
|
|
Rufinamide: MOA, Clinical Indications
|
Mechanism of action:
-Blocks the Na+ channel; prolonging inactive state Clinical Indications -Adjunctive treatment for seizures associated with Lennox Gastaut syndrome -Refractory partial seizures |
|
|
What is the standout AE for Rufinamide?
|
shortens QT interval
|
|
|
Ezogabine: MOA, Clinical Indications
|
Mechanism of action:
-Opens KCNQ2/3 voltage-gated potassium channels, activating M-current, which regulates neuronal excitability and suppresses epileptic activity Clinical Indications: -Adjunct in partial Seizures |
|
|
What is the standout AE for Ezogabine?
|
urinary retention
|
|
|
Generalized Seizures
|
Ethosuximide
-Phensuximide -Methsuximide Valproic Acid/Sodium Valproate |
|
|
Ethosuximide
|
Mechanism of action: Inhibits the Ca2+ currents of -T-type currents
T-type Ca2+ channels provide pacemaker currents in thalamus neurons --Generate the rhythmic cortical discharge of an absence attack Clinical Indication: -Absence (DOC) |
|
|
What's the point of Methsuximide and Phensuximide?
|
I don't know.
Methsuximide more toxic than ethosuximide Phensuximide less effective than ethosuximide |
|
|
Valproic Acid & Sodium Valproate
|
Mechanism of Action
-Blocks high frequency neuronal firing --Blockade of Na+ currents --Blockade of NMDA receptor mediated excitation --GABA levels are increased after valproic acid ---Enhanced synthesis or inhibited clearance mechanisms |
|
|
Valproic Acid & Sodium Valproate: Clinical Use
|
Absence seizures in the presence of generalized tonic-clonic attacks
Myoclonic seizures Simple partial Complex partial Partial with secondarily generalized tonic-clonic Tonic-clonic Bipolar disorder Migraine prophylaxis |
|
|
Valproic Acid & Sodium Valproate: Drug Interactions
|
Displaces phenytoin from plasma proteins
Inhibits the metabolism of phenobarbital, phenytoin, carbamazepine Decreases the clearance of lamotrigine |
|
|
What are the 3 black box warning for Valproic acid and Valproate?
|
Hepatotoxicity is severe adverse reaction
-Greatest risk for young patients (<2 yrs) or patients taking multiple drugs -Treat with oral or IV L-carnitine -Can be reversible if caught in time Pancreatitis -Increased risk of spina bifida in the offspring of women taking drug during pregnancy |
|
|
Benzodiazepines MOA
|
Increase GABA inhibition but do not work AS GABA
-Bind to allosteric site on GABAA receptor |
|
|
Diazepam and Lorazepam
|
Diazepam
-Given IV or rectally; can be given orally for long-term treatment -Stops continuous seizure activity Lorazepam also treats continuous seizure activity |
|
|
Clonazepam
|
Long-acting; treats absence seizures, myoclonic seizures
Adverse Effects: sedation |
|
|
Clorazepate dipotassium
|
Adjunct to treat partial seizures in adults
Adverse Effects: drowsiness and lethargy |
|
|
What is given as adjunct in patients >2 yo with Lennox-Gastaut?
|
Clobazam
|
|
|
Acetazolamide is an inhibitor of ______.
|
Mechanism of action:
-Inhibitor of carbonic anhydrase --Possibly causes mild acidosis in the brain --Possibly causes diminished release of bicarbonate ions from GABAergic neurons Clinical Indications: all seizure types -Tolerance quickly develops |
|
|
When does vagal nerve stimulation work best?
|
Works best for patients with REFRACTORY partial seizures
-Or when seizure meds are poorly tolerated Goal -To prevent seizures by regularly sending electrical pulses to the vagus -May prevent seizures (if patient senses one coming) --Move magnet over battery |
|
|
Status Epilepticus
|
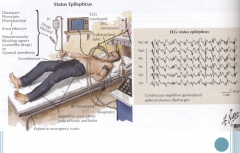
Recurrent episodes of tonic-clonic seizures
-Remain unconscious and without normal muscle movement between episodes -Lack of oxygen can lead to brain damage Requires immediate cardiovascular, respiratory, and metabolic management |
|
|
In the treatment of satus epilepticus, what goes into the 1st and 2nd IVs? What do you give if no response from the 2nd?
|
First: IV Diazepam or Lorazepam (30-45 min. seizure free)
Second: IV Phenytoin (push or saline; not diluted in glucose) -Need to monitor cardiac rhythm and BP in elderly --Cardiotoxicity d/t propylene glycol (dissolving agent) IV Fosphenytoin -Freely soluble in IV solutions (safer) -More potent than Phenytoin Phenobarbital – no response to phenytoin -Adverse Effects: respiratory depression |
|
|
What drugs require non-hormonal methods of birth control?
|
Use non-hormonal methods of birth control with these drugs:
Phenytoin Phenobarbital Carbamazepine Topiramate Oxcarbazepine Felbamate Increased seizure frequency at time of menses |
|
|
AED teratogenecity: Phenytoin – fetal hydantoin syndrome
|
“Symptoms in affected individuals may include abnormalities of the skull and facial features, growth deficiencies, underdeveloped nails of the fingers and toes, and/or mild developmental delays. Other findings occasionally associated with this syndrome include cleft lip and palate, having an abnormally small head (microcephaly) and brain malformations with more significant developmental delays.[1] “
|
|
|
What two other AEDs are linked to fetal hydantoin syndrome?
|
Linked to phenobarbital and carbamazepine
|
|
|
What risk is associated with Valrpoate? Topiramate?
|
Valproate – spina bifida (risk is 1-2%)
Topiramate – human male (hypospadias) |
|
|
Withdrawal
|
Patient must be seizure-free for several years
Can cause increase frequency and severity Benzodiazepines and barbiturates are difficult to discontinue – may take weeks to months |
|
|
What's wrong with generic AEDs?
|
Has become evident that generic drugs may not be as therapeutically stable as brand drugs
-“range” for the dose of active ingredient Increased risk of breakthrough seizures |
|
|
Don;t forget to look at slide 66
|
slide 66
|

