![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
3 Cards in this Set
- Front
- Back

Graph k1/k2=2 |
If k1/k2=2, k1 is 2× more than k2. The concentration of B is twice as much as C because the rate of formation from A-B is greater than A-C. @ equilibrium, the concentration of B will be twice as much as C |
|
|
Consecutive RXN'S (formulate rate law of I) |
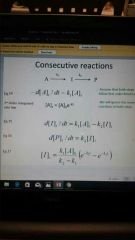
|
|
|
State special cases( limiting cases). Derive integrated rate law for (P) |

|

