![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
99 Cards in this Set
- Front
- Back
|
Polymer |
Long Chain of Monomers |
|
|
Homopolymers |
Polymers made from a singe type of monomer a-a-a-a-a-a-a |
|
|
Copolymers |
Polymers made from two types of monomers |
|
|
Random copolymer |
a-a-b-a-b-b-b-b-a- |
|
|
Alternating copolymer |
a-b-a-b-a |
|
|
Block(diblock) copolymer |
aaaaaaabbbbbbb |
|
|
Block(triblock) copolymer |
aaaaaaabbbbbbbaaaaaaaa |
|
|
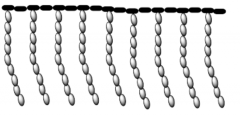
Graft copolymer |
|
|
|
Linear polymer |

|
|
|
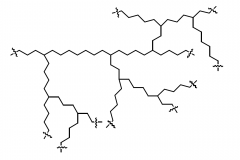
Branched polymer |
|
|
|
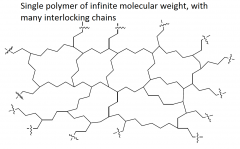
Network polymer |
|
|
|
Solubility of Linear polymer? |
Usually soluble |
|
|
Solubility of branched polymer? |
Usually soluble |
|
|
Solubility of Network polymer? |
May swell but will not dissolve |
|
|
Elastomers |

|
|
|
Thermosets |

|
|
|
Thermoplastics |

|
|
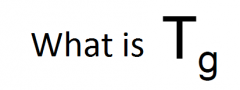
|
Glass transition temperature |
|
|
Amorphous Thermoplastics |

|
|
|
Semi-crystalline thermoplastics(SCT) |
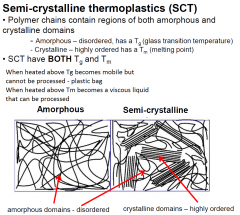
|
|
|
Molar Mass (M)(polymer context) +equation |
mass of 1 mole of polymer chains gmol⁻¹ M = xM₀ or M = nM₀ |
|
|
Degree of Polymerisation, (x or n)
|
Number of monomer per polymer chain
|
|
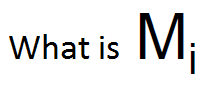
|
Molar mass of a polymer chain with i repeat units
|
|

|
Total number of polymer molecules of molar mass Mi
|
|

|

|
|
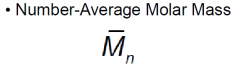
|
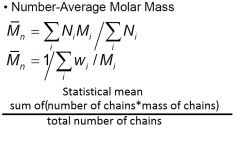
|
|
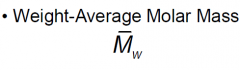
|
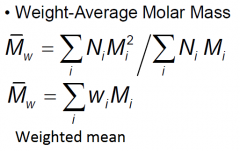
|
|
|
Polydispersity index (PDI)
|
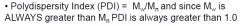
|
|
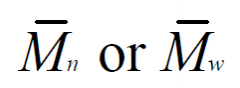
|

|
|
|
What do you understand by molecular weight distribution?
|
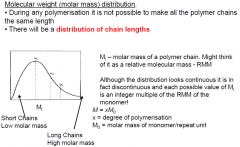
|
|
|
What do Mn and Mw mean relative to each other?
|

|
|
|
What is step growth polymerisation?
|

|
|
|
What is chain growth polymerisation?
|

|
|
|
What is a Polyester?
|
Polymer with units joined by ester groups, typically formed by the condensation (elimination)reaction between an diacid and a dialcohol or compound with one acid and one alcohol |
|
|
What is a Polycondensation reaction?
|
A Step growth polymeraisation reaction involving the condensation (elimination reaction) between monomers (acid and alcohol)
|
|
|
What is polyaddition?
|
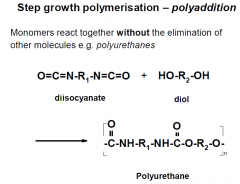
|
|
|
What is Carothers theory?(full, can ignore)
|

|
|
|
What is Carothers theory valid for?
|
Valid for RA₂ + RB₂ polymerisation in which there are equal numbers of A and B functional groups
|
|
|
Inplications of Carothers theory?
|
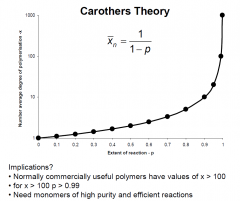
|
|
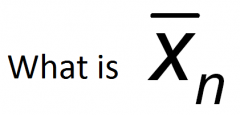
|
Number average degree of polymerisation
|
|
|
What is the number average degree of polymerisation? (equation)
|
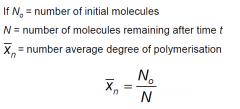
|
|
|
What is p?
|
Extent of reaction.
|
|
|
What is extent of reaction? (equation)
|
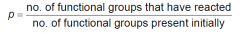
|
|
|
What is Carothers Theory?
|
Method of anaylsis for predicting the molar mass of polymers prepared by step growth polymerisation
|
|
|
Carothers Equation?
|
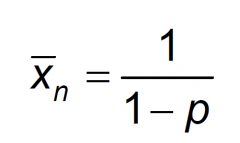
|
|
|
How is number average molar mass related to degree of polymerisation?
|
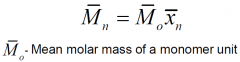
|
|
|
How do we get the mean molar mass of a monomer unit?
|

|
|
|
What is Carothers theory restricted to? |

|
|
|
What allows the prediction of size distributions?
|
Statistical theory(Flory)
|
|
|
What is statistical theory?
|

|
|
|
What does statistical theory give us?
|
allows us to predict the most probably weight fraction distribution at various extents of reaction p
|
|
|
Discuss Chain growth mechanisms?
|
describe polymerisations in which the polymer chain grows only by reaction between monomer and a reactive end group at the growing chain end. |
|
|
Steps of Chain growth?
|
initiation, propagation and termination |
|
|
Types of chain growth mechanisms?
|
controlled free radical (ATRP, RAFT, NMP) Anionic polymerisation cationic polymerisation ring opening polymerisation coordination polymerisation ring opening metathesis polymerisation |
|
|
Discuss initiation?
|
i) Formation of free radicals from an initiator by homolytic scission of a single bond (heat or light) or single electron transfer (redox) ii) addition of initiating radical to monomer |
|
|
What is the general structure of of monomers polymerised by free radical polymerisation?
|
CH₂=CR₁R₂ |
|
|
How is homolysis achieved?
|
Thermolysis occurs at 50-100⁰C, Photolysis, or single electron transfer is used when low temp required |
|
|
Discuss single electron transfer?
|
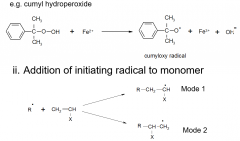
|
|
|
Which mode of single electron transfer dominates?
|
depends on steric hindrance, and the stability of the resulting radical
|
|
|
Propagation?
|
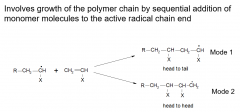
|
|
|
Termination?
|
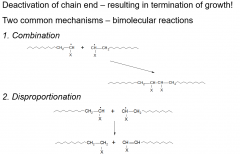
|
|
|
discuss chain transfer?
|
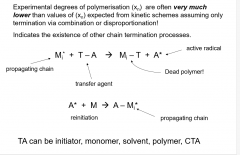
|
|
|
Discuss transfer constants?
|
transfer constants increase with ease of bond cleavage and stability of radical |
|
|
Discuss autoacceleration?
|
AKA gel effect occurs when conc of monomer is high, -during reaction viscosity increases =>reduced mobility of chains =>termination becomes diffusion controlled - kt decreases => propagation unaffected - monomer molecules are mobile + polymerisation is exothermic => speeds up reaction + viscocity makes dissipation of energy difficult => explosion |
|
|
Discuss Living (Anionic) Polymerisation?
|
-no inherant termination reaction |
|
|
Free radical v anionic polymerisation?
|

|
|
|
Experimental criteria for living polymerisation?
|
1) constant number of active centres 2) polymerisation proceeds until all monomer has been consumed 3) Mn is a linear function of conversion 4)Mn controlled by the stoichiometry 5) Narrow molecular weight distribution 6) block copolymers by sequential addition of monomers 7) chain-end functionalization by controlled termination is quantitative |
|
|
Why is Mn controlled by the stoichiometry of reaction for living polymerisation?
|

|
|
|
Why have living polymerisations got narrow molecular weight distribution?
|
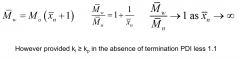
|
|
|
Random coils?
|
-statistical distribution of shapes for all the polymer chains instead of a single specific shape -polymer chains are dynamic -polymer chains in solution or in the amorphous regions of solid state assume (approximate) random coils |
|
|
Rod?
|
as the back bone of a coil becomes stiffer it will be come a rod |
|
|
Contour lengeth?
|
- length of the polymer chain along its back bone contour length = nL where n = number of bonds and l = length of bond |
|
|
Fully extended end to end distance?
|
nl cos(90-θ/2) |
|
|
Root mean square end-to-end distance?
|

|
|
|
Freely jointed chain model
|
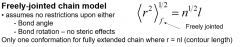
|
|
|
RMS vs contour length?
|
RMS is a factor of n¹/² smaller |
|
|
RMS radius of gyration?
|

|
|
|
Valence angle model?
|

|
|
|
Isomeric state theory?
|

|
|
|
Chain stiffness/ characteristic ratio?
|

|
|
|
Excluded volume?
|

|
|
|
Environmental effects upon polymer?
|

|
|
|
solvent effects on polymer?
|
In dilute solution there are no polymer-polymer interactions - in a good solvent, favourable solvent-polymer interactions => α > 1 = > polymer expands from unperturbed dimensios -bad solvent => α≤ 1 => polymer collapes often precipitating |
|
|
Theta conditions?
|
When polymer-solvent interactions are suffcient to cancel polymer segment - polymer segment interactions α = 1 solvent is called a theta solvent solvents can be good, bad or theta depending on temp |
|
|
T>Tg
|
-individual chains posses enough thermal energy to flow past each other -polymer has a high degree of free volume |
|
|
T<Tg
|
-polymer has much reduced free volume => no space to move -polymer becomes stiff and glassy |
|
|
what physical properties change when a polymer passes through Tg
|
Mechanical properties (solid to liquid) refractive index heat capacity thermal expansion |
|
|
First order and second order transitions?
|
second order transition - change in a first derivative e.g. Cp =(dH/dT), α = 1/V (dV/dT) α = volume thermal expansion coefficient |
|
|
Specific Volume and free volume around Tg
|
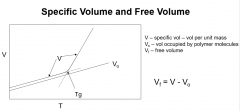
`
|
|
|
fractional free volume?
|
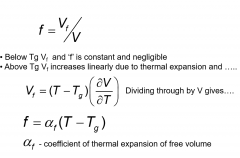
|
|
|
Dependence of Tg upon chemical structure?
|
1) flexible backbones give low Tg (unhindered rotation) 2) polymers with bulk side groups inclease Tg by restricting bond rotation => stiff chain 3) polymers with flexible side groups decrease Tg by increasing free volume 4) polymers with polar side groups increase Tg due to increased interactions 5) block and graft copolymers have multiple distinct values of Tg, while random / altenating have one Tg |
|
|
Dependence of Tg upon molar mass?
|

|
|
|
methods of measuring Tg?
|
Specific Volume vs T, Heat capacity ( DSC), mechanical properties DMA |
|
|
Differential scanning calorimetry? |
Two heaters heat two pans at the same heating rate, sample pan has to work harder, DSC measures how much more heat is required plot difference v Temp,
|
|
|
Dynamic mechanical analysis?
|
mearuser variation in mechanical properties as a function of force, strain, frequency, time and temperature (applying a constant fonce and varying T allows Tg to be measured (more sensitive than DSC) |
|
|
Discuss Thermoplastic elastomers?
|
- can be stretched, -recover -posses crosslinks at elevated temp:- thermoplastics -tehy can be heated and processed -cooled and then reheated |
|
|
explain thermoplastic elastomers?
|
crosslinks are not permanent covalent bonds but reversible physical crosslinks |
|
|
Phase separation of polymers?
|
polymer blends -macrophase separation upon cooling from melt block copolymers - microphase separation on cooling |
|
|
methods of multiblock synthesis?
|
sequential addition of monomers sequential additino of monomers difunctional initiator |

