![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
70 Cards in this Set
- Front
- Back
|
Atoms that gain or lose electrons and become electrically charged are called...
|
ions
|
|
|
A covalent bond in which electrons are not shared equally resulting in a molecule whose shape gives an uneven distrubition of charges
|
polar
|
|
|
The attraction of the positive hydrogen end of a polar molecule to the negative nitrogen or oxygen end of another polare molecule is called a...
|
hydrogen bond
|
|
|
2 or more atoms may combine to form a...
|
Molecule
|
|
|
If atoms of different elements combine, they form...
|
compound
|
|
|
Describe two ways that atoms may combine with other atoms?
|
Ionically or Covalently
|
|
|
Shorthand used to depict the numbers and types of atoms in a molecule
|
Molecular Formula
|
|
|
Illustration with lines connecting molecules together
|
Structural Formula
|
|
|
What's the difference between a polar molecule and an ion
|
A polar molecule is a covalent bond with in which electrons are not shared equally.
|
|
|
A polar molecule has an ______ number of electrons. (equal or unequal)
|
Unequal
|
|
|
What are some properties of Water
|
surface tension, solvent, Hydrogen bonds for a lattice structure when frozen, transport, cohesive
|
|
|
what are some technologies that utilize the polar nature of water
|
Ultrasound and MRI
|
|
|
What does a pH scale measure?
|
Hydrogen ion concentration
|
|
|
Chemicals that resisist pH change are called
|
Buffers
|
|
|
Substance formed by the reaction between an acid and a base
|
Salt
|
|
|
Substance that releases H+ ions
|
Acid
|
|
|
Substance that accepts H+ ions
|
Base
|
|
|
Chemical compounds that contain Carbon and Hydrogen
|
Organic
|
|
|
Substances that usually dissociate in water, forming ions
|
inorganic compounds
|
|
|
A substance that completely dissolves in water
|
Electrolyte
|
|
|
A substance that partially or doesn't dissolve in water
|
Non-Electrolyte
|
|
|
The maintenance of ions within the body in certain concentrations
|
Electrolyte Balance
|
|
|
These provide much of the energy that cells require and are made of Carbon, Hydrogen, and Oxygen
|
Carbohydrates
|
|
|
Glucose and Starch are examples of...
|
Carbohydrates
|
|
|
Group of organic chemicals that are insoluble in water, but soluble in organic solvents such as ether and cholorform?
|
Lipids
|
|
|
Single C-C bonds link all the carbon atoms are called _______ fatty acid
|
Saturated
|
|
|
Structural materials, energy sources and chemical messengers (hormones)
|
Proteins
|
|
|
Proteins play vital roles in metabolism as _____
|
enzymes
|
|
|
What key element does protein always include
|
Nitrogen (Carbon, Hydrogen, and Oxygen and sometimes Sulfur)
|
|
|
What are the building blocks of proteins
|
Amino Acis
|
|
|
What are the building blocks of Lipids
|
Glycerol, fatty acids, and phosphate groups
|
|
|
What are the building blocks of carbohydrates
|
Sugars
|
|
|
What are the building blocks of Nucleic Acids
|
Nucleotides
|
|
|
Tryglycerides, phospholipids, and steroids are examples of...
|
Lipids
|
|
|
Peptides are examples of...
|
Proteins
|
|
|
RNA and DNA are examples of
|
Nucleic Acids
|
|
|
Name the major parts of a cell?
|
Nucleus, Cytoplasm, adn Cell Membrane
|
|
|
This contains the genetic material (DNA) which directs the cell's functions
|
Nucleus
|
|
|
Composed of specialized structurs called cytoplasmic organelles suspended in a liquid called cytosol
|
Cytoplasm
|
|
|
Loosely coiled fibers containing protein and DNA within the nucleus
|
Chromatin
|
|
|
Location of ATP production from digested food molecules
|
Mitochondrion
|
|
|
Small RNA-containing particles for the synthesis of proteins
|
Ribosome
|
|
|
Membranous sac formed by the pinching off of pieces of cell membrane
|
Vesicle (Vacoule)
|
|
|
Dense body of RNA within the nucleus
|
Nucleolus
|
|
|
Slender tubes that provide movement in cilia and flagella
|
Microtubule
|
|
|
Organelles composed of membrane-bound sacs, canals, and vesicles for tubular transport
|
Endoplasmic Reticulum
|
|
|
Occupies space between the cell membrane and nucleus
|
Cytoplasm
|
|
|
Flattened membranous sacs that package a secretion
|
Golgi apparatus
|
|
|
Membranous sac that contains digestive enzymes
|
Lysosome
|
|
|
Separates nuclear contents from cytoplasm
|
Nuclear envelope
|
|
|
Spherical organelle that contains chromatin and nucleolus
|
Nucleus
|
|
|
Mainly composed of lipids and proteins
|
Cell Membrane
|
|
|
Membrane protein that receives and transmits messages into a cell
|
Receptor Protein
|
|
|
Membrane protein that forms pores, channels and carries in cell membrane, dransduce signals
|
Integral proteins
|
|
|
Protein that enables certain cells to touch or bind
|
Cellular Adhesion molecules (CAMs)
|
|
|
A + B >>>>> AB
|
Synthesis Reaction: When 2 or more atoms bond to form a complex structure
|
|
|
AB >>>>> A + B
|
Decomposition Reaction: When a Bond breaks to form simpler molecules, atoms, or ions
|
|
|
AB + CD >>>>> AD + CB
|
Exchange Reaction: Parts of 2 different types of molecules as bonds are broken and new bonds are formed
|
|
|
A + B >>><<< AB
|
Reversible Reaction: The products can change back into the original reactant
|
|
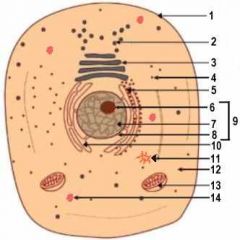
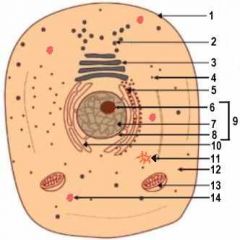
1
|
Cell Membrane
|
|
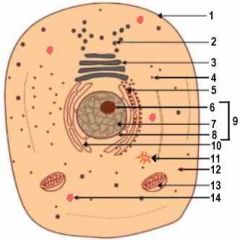
2
|
Vesicle (Secretory Structure)
|
|
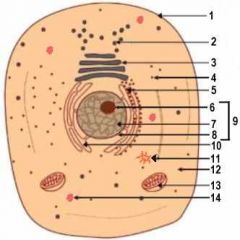
3
|
Golgi Apparatus
|
|
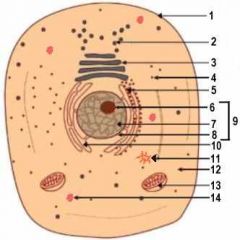
4
|
Ribosome
|
|
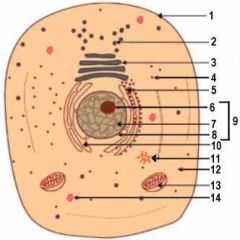
5
|
Rough ER
|
|
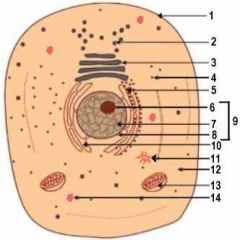
6
|
Nuclelous
|
|
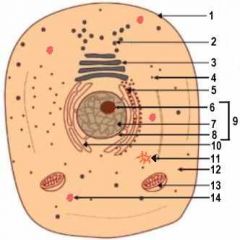
10
|
Smooth ER
|
|
|
11
|
Centresome
|
|
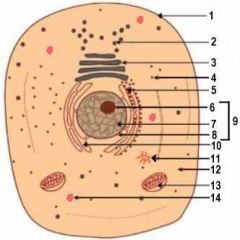
12
|
Cytoplasm
|
|
13
|
Mitochondrion
|
|
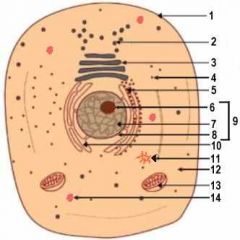
14
|
Lysosome
|

