![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
10 Cards in this Set
- Front
- Back
|
When a sample of gas is heated, it's thermal energy |
Increases |
|
|
The pressure exerted by a gas in a container depends on |
The number of collision between gas molecules and walls of container |
|
|
As atmospheric pressure decreases, the column of mercury in a barometer |
Deceases in heights |
|
|
Relationship between pressure of gas and absolute temperature |
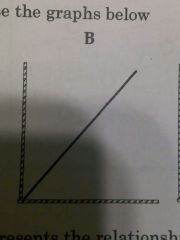
As temperature increases pressure increase |
|
|
Relationship between pressure of gas and volume |
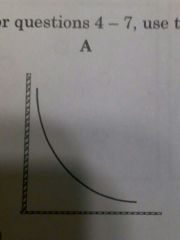
As volume decrease pressure increase |
|
|
Relationship between volume of gas and Celsius temperature |
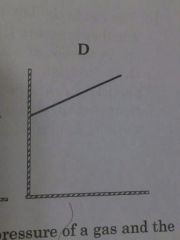
As temperature increases the volume increases but the temperature does not have to start out on zero |
|
|
What is going on at the molecular level when a thermometer is placed in warm water |
As the thermometer is placed in warm water the molecules spread out and move faster causing the temperature to rise |
|
|
Represent in wooshies more pressure |
More wooshies |
|
|
What happens when particles are added to a space |
The pressure of the gas would increases because as the number of particles increase the pressure increase |
|
|
Pg-Pa=Ha-Hg |
PG is the gas dome. HG is closet to the gas dome. PA is the top. HA is closet to the top |

