![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
284 Cards in this Set
- Front
- Back
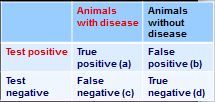
How do you calculate sensitivity from this 2x2 table?
|
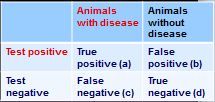
sensitivity - a/(a+c)
|
|
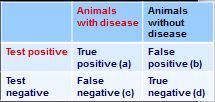
how do you calculate specificity from this 2x2 table?
|
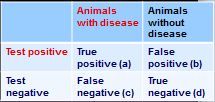
specificity = d/(b+d)
|
|
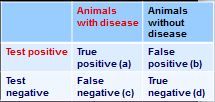
how do you calculate positive predictive value from this 2x2 table?
|
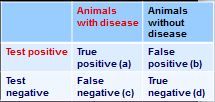
PVP = a/(a+b)
|
|
|
what is central to clinical decision making?
|
diagnosis
|
|
|
what are two basic types of tests collected from blood to diagnose viral infection? What type of samples should be collected for each?
|
1. virus isolation - EDTA/heparin; refrigerate before transport
2. serology - clotted blood; refrigerate, spin, separate serum |
|
|
fecal collection for viral diagnosis
- how much should you collect? - how should the sample be stored? |
- collect at least 10 grams of voided feces
- put in viral transport medium and refrigerate for transport |
|
|
what are the two types of materials used for rectal/cloacal swabs and what type of viral diagnostic test is each one indicated for?
|
1. dacron swab: virus isolation
2. cotton swab: PCR |
|
|
why shouldn't you use cotton swabs for virus isolation, and instead use dacron?
|
because cotton is bleached and residual bleach can lyse the viruses
|
|
|
what are common sites of collection for virus diagnosis from:
- bird skin? - eye? |
- feather follicles
- conjuntival swab in VTM - corneal imprints on glass slides - tears |
|
|
how are vesicles submitted to virology labs?
|
≈2 grams of vesicular epithelium in transport medium
|
|
|
what are the four notifiable vesicular diseases?
|
1. Foot and Mouth disease (picaronavirus of pigs, cattle, sheep)
2. Vesicular stomatitis (rhabdovirus of horses, cattle, and pigs) 3. vesicular exanthema (calicivirus of swine) 4. swine vesicular disease (enterovirus of swine) |
|
|
what is the best way to collect samples of discharges for a virology lab in
- the vagina? - semen? - saliva, tears, nasal? - milk? |
- the vagina: vaginal/preputial washing/swabs
- semen: AV/penis extrusion - saliva, tears, nasal: cotton, dacron, or gauze; soak for 1 minute; transport at 4 °C in VTM - milk: collect second stream |
|
|
when storing histopath samples for viral analysis, what should you not do with them?
|
freeze
|
|
|
what is the difference between direct examination and indirect examination with regards to a virus diagnostic test?
|
- direct examination: the actual virus or something produced by the virus
- indirect examination: something affected by, not not directly produced by the virus |
|
|
what are six indicators of current viral infection?
|
1. isolation of agent
2. ID of the agent's genes 3. clinical signs 4. pathognomonic signs 5. biochemical changes 6. demonstration of an immune response |
|
|
what are three indicators of past viral infection?
|
1. clinical history
2. pathognomonic changes 3. demonstration of antibodies |
|
|
what are two techniques used for viral antigen detection?
|
1. IFA
2. ELISA |
|
|
how is light microscopy used to perform direct examination of a viral infection?
|
1. histological appearance
2. inclusion bodies |
|
|
name some observations used in indirect examination of a viral infection
|
- cytopathic effects
- hemadsorption - embryo: lesions, pocks on chorioallantoic membrane - susceptible animals: clinical signs and lesions; virological and serological ID methods |
|
|
which epidemiological parameter tells you the probability that an animal showing a positive test result actually has the disease?
|
positive predictive value
|
|
|
which epidemiological parameter tells you the probablility that a negative test result is a true negative?
|
negative predictive value
|
|
|
what does sensitivity tell you about a test?
|
the probability that a truly diseased animal will not have a false negative
|
|
|
what does specificity tell you about a test?
|
the probability that an animal without disease will not register as a false positive
|
|
|
which epidemiological parameter tells you the probability that a truly diseased animal will not have a false negative
|
sensitivity
|
|
|
which epidemiological parameter tells you the probability that an animal without disease will not register as a false positive
|
specificity
|
|
|
what does positive predictive value tell you about a test?
|
the probability that an animal showing a positive test result actually has the disease?
|
|
|
what does negative predictive value tell you about a test?
|
the probability that a negative test result is a true negative (or not a false-negative)
|
|
|
normal and abnormal test results will have overlapping bell curves. What are the two extremes in determining the cut-off point of these tests and what are their advatnages and disadvantages?
|
1. any positive test value is a positive (maximum sensitivity): few false negatives but many false positives
2. any negative test result is considered a negative (maximum specificity): few false positives but many false negatives |
|
|
the probablility of a random animal being positive is called what?
|
prevalance
|
|
|
the proportion of animals given a positive test result is called what?
|
apparent prevalance
|
|
|
the actual proportion of infected animals is called what?
|
true prevalence
|
|
|
if prevalence is low, what epidemiological parameters of a test are the most important.
|
you want high specificity to avoid false-positives
|
|
|
what epidemiological parameter of a diagnostic test is the most important in ruling out a disease?
|
sensitivity; you want very few false negatives
|
|
|
regarding sensitivity and specificity, what are the rules of thumb to
- rule out a disease -rule in a disease |
- rule out: ≥ 95% sensitivity and ≥ 75% specificity
- rule in: ≥75% sensitivity and ≥ 95% specificity |
|
|
if you have a positive FeLV SNAP test, what are three recommendations?
|
1. retest with a confirmatory test such as IFA or virus isolation
2. isolate and retest after 4 weeks 3. if negative second test, declare negative if another negative test 8 weeks later |
|
|
porcine parvovirus:
- nucleic acid - envelope? - geographical distribution |
- DNA
- non-enveloped - worldwide distribution |
|
|
what does SMEDI stand for?
|
Still birth
Mummification Embryonic Death Infertility |
|
|
what are the clinical signs of porcine parvovirus in the sow?
|
none - they can be carriers
|
|
|
what aspect of the porcine parvirus makes them "want" to propagate in the fetus?
|
parvoviruses target rapidly dividing cells
|
|
|
what are four factors that may influence the outcome of a porcine parvovirus infection?
|
1. immunocompetence
2. gestational age 3. period of infection 4. pathogenicity of the virus strain |
|
|
what are the outcomes of porcine parvovirus infection in utero:
-of an early infection? - infection at 50-60 days of gestation? - infection at 72+ days of gestation |
- early: abortion, absorption of fetuses, usually no mummification
- 50-60 days: abortion, mummification - 72+ days: survival possilbe |
|
|
when does a pig fetus acquire immunocompetence to porcine parvovirus infection?
|
60-70 days of gestation
|
|
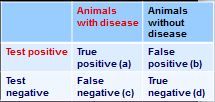
how do you calculate negative predictive value from this 2x2 table?
|
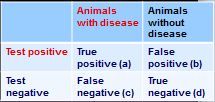
PPN = d/(c+d)
|
|
|
what are four families of DNA viruses that commonly infect swine?
|
1. Parvoviridae
2. herpesviridae 3. Poxviridae 4. circoviridae |
|
|
if a group of sows is aborting mummified fetuses at various stages of pregnancy, what infection should you suspect?
|
porcine parvovirus
|
|
|
describe the histopathology of porcine parvovirus in an aborted fetus
|
- perivascular cuffing around veins and capillaries in the brain
- meningoencephalitis - interstitial hepatitis - nephritis |
|
|
what three things can give you a presumptive diagnosis of porcine parvovirus?
|
1. clinical signs (e.g. mummified fetuses)
2. epidemiology 3. necropsy |
|
|
what is an important differential diagnosis for porcine parvovirus?
|
SMEDI
|
|
|
what are two tests to definitively diagnose porcine parvovirus?
|
1. antigen by IFA (cryostat sections of tissues)
2. virus isolation in susceptible cells |
|
|
how is porcine parvovirus prevented and controlled?
|
- routine vaccination of sows and gilts with MLV vaccine; this prevents the virus from passing through the placenta
- piglets are generally NOT vaccinated - very difficult virus to eliminate |
|
|
to what family of viruses does pseudorabies virus belong?
|
herpesviridae
|
|
|
what are the two named diseases that are caused by pseudorabies?
|
1. Aujeszky's Disease
2. Mad itch |
|
|
what are the reservoir hosts for the pseudorabies virus?
|
enzootic in swine, and some other non-swine species
|
|
|
how is pseudorabies virus transmitted?
|
the oral-nasal route: breathing, feeding, or social activity
|
|
|
what is the most important organ system and tissues infected by pseudorabies virus?
|
CNS and nerve tracts
|
|
|
what are six clinical signs of pseudorabies virus infection in swine?
|
1. interstitial pneumonia
2. fever 3. excessive salivation 4. depression 5. tremors 6. convulsions |
|
|
what are the main clinical signs of sows infected with pseudorabies?
|
SMEDI
|
|
|
describe the outcome of intrauterine infection in the sow with pseudorabies:
- at the first 30 days - at the early 2nd trimester - at term |
- at the first 30 days: abortion, absorption, low farrowing rate
- at the early 2nd trimester: abortion, fetus expelled - at term: pigs born dead, macerated, or live-infected |
|
|
besides intrauterine transmission, what is another route of vertical transmission of pseudorabies?
|
colostrum
|
|
|
comment on the latency and recrudescence of pseudorabies in pigs
|
(swine herpesvirus-1)
- recovered pigs are immune but remain latently infected (inactive in sensory ganglia of brain or CNS) - under traumatic or stressful conditions, the virus will recrudesce and shed without clinical signs |
|
|
if pseudorabies recrudesces and does show clinical signs, what will you see?
|
herpetic vesicles in the snout and face, and raw areas on the ears and shoulders
|
|
|
comment on pseudorabies in non-swine species
- morbidity and mortality - common clinical sign - how is it transmitted? |
- rare, but usually fatal
- severe itching, followed by rabies-like clinical signs (mania, encephalitis, paralysis, death) - transmission is usually from presence of infected swine or bites from infected rats |
|
|
what are the atypical gross lesions associated with pseudorabies infection?
|
1. edema and necrosis at infection site
2. congestion in the nasal mucosa and pharynx 3. congestion, edema, and hemorrhages in lungs 4. hemorrhages in kidneys and pericardium 5. slight splenomegaly and small necrotic foci in the spleen and liver |
|
|
a steer presenting for aggression and gaping wounds from scratching against trees and the fence might have what?
|
pseudorabies
|
|
|
how is pseudorabies infection definitively diagnosed?
|
- viral antigen by FA
- serology: paired sera with a 4-fold increase of titers - virus isolation in susceptible cells |
|
|
comment on the vaccines available for pseudorabies.
|
- MLV: effective in reducing death losses in endemic areas, but not effective in reducing infection, re-infection, latent infection, or carrier status
- inactivated: efficacious and mostly used to vaccinate sows to provide antibodies in the colostrum for newborns |
|
|
why is use of MLV for vaccination of pseudorabies controversial?
|
because there are test-and-slaughter eradication efforts in the US. MLV can interfere with testing and may establish a non-pathogenic latent infection in the vaccinated host.
|
|
|
how long should you quarantine new swine to reduce the risk of exposure of your naïve population to a virus such as pseudorabies?
|
at least 12 days
|
|
|
if a pseudorabies outbreak occurs on a farm, how long after clinical signs cease should the farmer wait to export animals from the premises?
|
3 weeks
|
|
|
what is another name for porcine cytomegalovirus? What is the common name for the disease it causes?
|
- porcine herpesvirus-2
- inclusion body rhinitis |
|
|
what tissues are infected with porcine cytomegalovirus?
|
the bones
|
|
|
who is most susceptible to porcine cytomegalovirus?
|
(porcine herpesvirus-2)
young pigs < 2 weeks old (pigs over 4 months old show no clinical signs) |
|
|
what viral infection can predispose a piglet to atrophic rhinitis or Glasser's disease?
|
porcine cytomegalovirus
|
|
|
comment on the prevention/control of porcine cytomegalovirus
|
- good hygiene
- no vaccine available |
|
|
what are two important Poxviridae that infect swine? Which one is zoonotic?
|
1. swinepox (swine only)
2. vaccinia virus (zoonotic) |
|
|
a group of piglets presents with an acute onset of fever, followed by small papules on the abdomen and inner thighs? What are two differentials?
|
1. swinepox
2. vaccinia virus |
|
|
how are vaccinia and swinepox diagnosed presumptively? definitely?
|
- presumptive: clinical signs and lesions
- lab: inoculation of chicken embryos or cell culture |
|
|
how are vaccinia and swinepox prevented and controlled?
|
- no vaccine
- good hygiene - lice control |
|
|
what arthropod vector is known to spread vaccinia and swinepox?
|
lice
|
|
|
porcine circovirus:
- genome - envelope? - symmetry |
- circular ssDNA
- non-enveloped - icosahedral |
|
|
what are the two types of porcine circoviruses and comment on their pathogenicity?
|
- PCV-1 is non-pathogenic
- PCV-2 causes postweaning multisystemic wasting syndrome (PMWS, now known as porcine circovirus associated disease, PCVAD) and is the most economically important porcine viral disease in the world |
|
|
what is PMWS and what virus causes it?
|
- postweaning multisystemic wasting syndrome
- caused by porcine circovirus-2 |
|
|
PMWS:
- most susceptible animals - morbidity - mortality - immune status of infected animals |
- low morbidity
- high mortality - affects high health herds |
|
|
what are four clinical signs for PMWS?
|
(after weaning)
1. progressive weight loss 2. dyspnea 3. enlarged lymph nodes 4. jaundice |
|
|
what are three gross pathological lesions associated with PMWS?
|
1. markedly enlarged lymph nodes***
2. non-collapsed, tan-mottled lungs 3. tan-colored, swollen liver |
|
|
markedly enlarged inguinal lymph nodes found in PM of a 12-week weanling piglet might be which disease (and associated virus)?
|
- PMWS
- causes by porcine circovirus-2 |
|
|
what are some histopathological findings of the lymph nodes with PMWS?
|
- lymphoid depletion
- histiocytic replacement of follicles in lymphoid tissues (granulomatous lymphadenitis) |
|
|
what are two routes of transmission of porcine circovirus?
|
- horizontal: most frequently via the oronasal route
- vertically: transplacental |
|
|
how is porcine circovirus prevented and controlled
|
- herd management: segregated early weaning; all-in all-out
- vaccinating piglets (@ 4 weeks) is very effective - vaccinating sows unknown |
|
|
what porcine viruses cause SMEDI?
|
1. porcine parvovirus
2. porcine enteroviruses 3. pseudorabies (PHV-1) |
|
|
how many serotypes of porcine enterovirus have been reported? Which ones cause disease?
|
- nine
- only PEV 1 and PEV 9 cause disease |
|
|
what disease is caused by
- porcine enterovirus-1? - porcine enterovirus-9? |
- PEV-1: polioencephalomyelitis
- PEV-9: swine vesicular disease |
|
|
what are the two important strains of porcine enterovirus-1? Which one is more virulent?
|
Teschen (more virulent) and Talfan
|
|
|
in what family of viruses are the porcine enteroviruses?
|
picornaviridae
|
|
|
what are the 3 stages of the disease process for porcine enterovirus-1 infection?
|
1. infection in the alimentary tract and associated lymph nodes
2. viremia 3. invasion of the CNS → polio |
|
|
what are the CNS signs of porcine enterovirus-1 infection?
|
- fever, anorexia, depression, tremors, convulsions, coma, and death
- mortality up to 70% |
|
|
what is the most important differential for porcine enterovirus-1? What are 3 others?
|
- pseudorabies
1. African swine fever 2. classical swine fever (hog cholera) 3. hemagglutinating encephalomyelitis |
|
|
how is porcine enterovirus-1 definitively diagnosed?
|
virus isolation from brain, intestinal tract, and feces
|
|
|
how is porcine enterovirus-1 prevented and controlled?
|
- there is no vaccine in the US
- import quarantine and restrictions |
|
|
what virus causes swine vesicular disease?
|
porcine enterovirus-9
|
|
|
what are three differentials for swine vesicular disease?
|
1. foot and mouth disease
2. vesicular stomatitis 3. vesicular exanthema |
|
|
what are the best samples for diagnosing an infection with porcine enterovirus-9?
|
vesicular fluids (note. PEV-9 causes swine vesicular disease)
|
|
|
how is porcine enterovirus-9 prevented and controlled?
|
(swine vesicular disease)
- no vaccine - restriction of SVDV spread: quarantine, movement restriction, test-and-slaughter |
|
|
porcine rotavirus:
- what family of viruses? - what disease does it cause? |
- Reoviridae
- causes young piglet diarrhea |
|
|
which pigs are most predisposed to porcine rotavirus?
|
piglets 10-28 days of age, when maternal antibodies have waned
|
|
|
what protects newborn piglets from porcine rotavirus?
|
IgA in the colostrum
|
|
|
how is porcine rotavirus acquired and how does it cause disease?
|
- acquired by ingestion (fecal-oral)
- replicates in the mature epithelial cells of the SI villi, causing them to shorten and blunt, thus causing severe diarrhea - it also potentiates enterotoxic E. coli |
|
|
why might you treat a rotaviral infection with antibiotics?
|
if there is a concurrent E. coli infection; ETEC is potentiated by rotaviral infection
|
|
|
how due piglets with a rotaviral infection usually die?
|
from dehydration; destruction of the intestinal villi → no water absorption
|
|
|
what are three ways to control and prevent porcine rotavirus?
|
1. commercially available bivalent vaccine for swine transmissible gastroenteritis virus and porcine rotavirus, to immunize the sows
2. good hygiene in farrowing and nursing facilities 3. lactogenic immunity. Make sure all piglets suckle early and continuously. |
|
|
what is the most important viral disease of young piglets? What type of virus is it? Envelope?
|
swine transmissible gastroenteritis virus (TGEV), a member of Coronaviridae, which is an enveloped virus
|
|
|
which swine viruses infect and destroy the intestinal villi?
|
1. porcine rotavirus
2. swine transmissible gastroenteritis virus (TGEV), a coronavirus |
|
|
comment on the seasonality of TGEV
|
since coronaviruses are enveloped, they are temperature sensitive and thus this virus usually occurs in the winter.
|
|
|
how is TGEV acquired?
|
- inhalation or ingestion
- large amounts of TGEV in the feces and respiratory secretions |
|
|
what is the most important means of spreading TGEV among piglets in farrowing and nursery facilities?
|
fecal shedding
|
|
|
a piglet with severe, watery and yellowish diarrhea with no blood may have which disease? What causes this type of diarrhea?
|
TGEV (coronavirus). Destruction of the intestinal villi makes them unable to digest milk or absorb water.
|
|
|
if TGEV outbreaks only occur in the winter, because they are enveloped coronaviruses, why can they keep recurring year after year?
|
because the virus survives in the lungs of sows without producing clinical signs
|
|
|
where specifically in the intestine does TGEV replicate?
|
in the TIP of mature epithelial cells of the SI villi (has no effect on the crypt (Lieberkuhn cells) are the base of the villi)
|
|
|
at PM, how is TGEV differentiated from E. coli?
|
with TGEV, the SI walls are very thin, distended, and transparent due to destruction of the villi. With E. coli, the villi are not affected
|
|
|
what are four differentials for TGEV in a sick piglet?
|
1. rotavirus diarrhea
2. colibacillosis 3. clostridial enteritis 4. coccidiosis |
|
|
what are three ways to prevent and control TGEV?
|
1. vaccinate sows to produce IgA in the colostrum
2. lactogenic immunity (IgA and IgG; note IgG does not survive GI enzymes): early and continuous suckling--every piglet 3. good hygiene |
|
|
why would you not actively immunize immunologically naïve newborns for TGEV?
|
because the disease will run its course before the immune system can develop antibodies.
|
|
|
what two diseases are causes by hemagglutinating encephalomyelitis virus?
|
1. vomiting and wasting disease
2. hemagglutinating encephalomyelitis |
|
|
vomiting and wasting disease:
- susceptible piglets - important clinical signs |
- suckling piglets 1-3 weeks
- vomiting, depression, dehydration, constipation |
|
|
though they all cause wasting and dehydration, what is an important distinction in clinical signs between hemagglutinating encephalomyelitis and TGEV/rotaviral infections in piglets?
|
TGEV and rotavirus cause diarrhea; HEV causes vomiting and constipation
|
|
|
how is hemagglutinating encephalomyelitis virus transmitted?
|
by nasal secretions
|
|
|
what are three important differentials for hemagglutinating encephalomyelitis virus?
|
1. polio (porcine enterovirus-1)
2. pseudorabies (porcine herpesvirus) 3. classical swine fever (Hog cholera) |
|
|
why does hemagglutinating encephalomyelitis virus cause dehydration in piglets?
|
from vomiting
|
|
|
what are the two most common swine influenza subtypes?
|
H1N1 and H3N2
|
|
|
swine influenza virus belongs to which family?
|
Orthomyxoviridae
|
|
|
swine influenza virus:
- seasonality - morbidity and mortality - transmission |
- most cases in autumn, winter, and early spring
- high morbidity, low mortality - transmission via aerosol droplets |
|
|
how can swine influenza virus, which is normally mild, cause much more severe disease (without mutating)?
|
is predisposes the animal to secondary bacterial infections such as Haemophilus somnus (aka Histophilus somni)
|
|
|
what are four clinical signs of swine influenza?
|
1. congestion in the URT
2. swelling of the cervical and mediastinal lymph nodes 3. exudate in the air passages 4. pneumonia, with consolidation of the lung |
|
|
what are some histopathological findings for swine influenza?
|
exudative changes in the bronchi, bronchioles, alveoli, atelectasis, and emphysema
|
|
|
what are three ways to prevent and control swine influenza virus?
|
1. early diagnosis and separation of animals (outbreaks are difficult to control)
2. ID of strain and sub-strain important for animal management and human health 3. vaccination |
|
|
economically, what does the USDA list as the #1 swine pathogen?
|
PRRS virus
|
|
|
PRRSV is in which family of viruses?
|
arteriviridae
|
|
|
what are the two basic syndromes caused by PRRSV?
|
1. reproductive failure in sows
2. respiratory disease (pneumonia) in growing pigs |
|
|
PRRSV:
- North America versus Europe - genome type - envelope? |
- N. America and Europe have PRRSV's with two distinct genotypes
- (+) sense RNA virus - enveloped |
|
|
in what three was is PRRSV transmitted?
|
1. pig movement and aerosols
2. direct contact and internasal route 3. semen from viremic boars |
|
|
what three reservoirs have been proposed for PRRSV (but debunked)?
|
1. avian species (i.e. mallard ducks)
2. vectors such as mosquitos (but virus does not replicate in mosquitos) 3. rodents, but rodents are not susceptible to PRRSV |
|
|
how long does PRRSV persist in larger herds?
|
up to 156 days
|
|
|
what are clinical signs of PRRSV in newborn or nursing piglets?
|
- respiratory signs
- "thumping" breathing sounds - abdominal breathing - pneumonia - occasionally CNS signs |
|
|
what are clinical sings of PRRSV in growing-finishing pigs?
|
- fever
- anorexia - pneumonia - listlessness - pruritis (rare) |
|
|
what are important clinical signs of PRRSV in breeding sows?
|
reproductive failures; SMEDI-like signs
|
|
|
what are clinical signs of PRRSV in boars?
|
- inappetence
- depression - fever - respiratory problems |
|
|
what is the proposed pathogenesis of PRRSV?
|
entry through nasal, tonsillar, and pulmonary macrophages → replication inside the macrophage → viremia → clinical signs
|
|
|
what are two major obstacles in control of the PRRS virus?
|
1. persistent, clinically normal infections (enzootic)
2. heterogeneity in the strains → difficult to develop an effective vaccine |
|
|
what are three gross pathological lesions associated with PRRSV?
|
1. rhinitis and loss of cilia on the nasal turbinates
2. tan colored lungs that fail to collapse 3. enlargement of lymph nodes |
|
|
why is detection of serum antibodies no longer indicated to diagnose PRRSV infection?
|
because it is an enzootic, widespread infection worldwide, and the use of MLV or killed vaccines (which do not work) causes false-positives.
|
|
|
why are current vaccines not effective against PRRSV?
|
- because there is so much heterogeneity in the numerous strains of the virus
- MLV vaccine virus reversion |
|
|
what type of virus causes vesicular exanthema of swine?
|
a calicivirus
|
|
|
what are three differentials for vesicular exanthema virus of swine?
|
1. foot and mouth disease
2. vesicular stomatitis 3. swine vesicular disease |
|
|
what is the other name for hog cholera virus? What type of virus is it?
|
- classical swine fever virus
- flavivirus |
|
|
what are the natural hosts of hog cholera virus?
|
only swine
|
|
|
what are six important clinical signs of pigs infected with hog cholera?
|
1. huddling and piling up as if they were cold
2. weakness, staggering, and tendency to sit like a dog 3. erythema, especially on the abdomen, flank, and legs 4. diarrhea 5. running movements (paddling) due to CNS 6. "goose stepping" (CNS) |
|
|
what are important gross pathological lesions of hog cholera in the:
- acute form? - chronic form? |
- acute: marginal infarcts on the spleen and gall bladder; raised button ulcers of the colon mucosa; enlarged lymph nodes, petechial hemorrhages in various organs
- chronic: necrotic ulcers in the large intestine; secondary bacterial pneumonia; enteritis |
|
|
what infection is impossible to distinguish from hog cholera without lab tests?
|
African swine fever virus
|
|
|
what are five differentials for hog cholera?
|
1. salmonellosis
2. acute erysipelas (ecchymotic hemorrhages, not petechiae) 3. acute pasteurellosis 4. viral encephalomyelitis 5. African swine fever |
|
|
how is hog cholera prevented and controlled?
|
- eradicated in the US - notifiable and reportable disease
- no vaccination in the US, but they do vaccinate in Europe |
|
|
African swine fever virus belongs to which family? Genome? Envelope?
|
- asfarviridae
- ds enveloped DNA virus |
|
|
what are the reservoir hosts and vectors for African swine fever virus?
|
- wild hogs are the reservoir hosts
- ticks are the vector |
|
|
comment on the morbidity and mortality of African swine fever virus
|
high morbidity and almost 100% mortality
|
|
|
what is the pathogenesis of African swine fever virus?
|
bite by ticks → replication in the lymph nodes → viremia → replication in lymphoid organs and vascular endothelium → widespread hemorrhaging
|
|
|
what are three differential diagnoses for African swine fever virus?
|
1. hog cholera
2. erysipelas 3. salmonellosis |
|
|
how is African swine fever prevented and controlled in the US?
|
- no vaccination in the US
- entire US swine population is susceptible - control of meat import and destruction of waste food from airplanes and ships from endemic countries - quarantine and slaughter to stop the disease |
|
|
describe the lesions caused by equine papillomavirus that was
- acquired - congenital |
- acquired: skin and genital mucosa lesions
- congenital: aural plaques |
|
|
what are two ways in which equine papillomavirus is transmitted?
|
1. direct contact
2. abrasions & wounds |
|
|
what does an equine papillomavirus infection on the nose and lips look like?
|
small, anchored papillomas
|
|
|
which virus causes equine genital warts?
|
Equine papillomavirus 2
|
|
|
a locally aggressive, non-metastatic, fibroblastic skin tumor of the horse
|
sarcoid
|
|
|
what causes sarcoids in the horse?
|
BPV 1, 2, and 5
|
|
|
in which breed of horse are sarcoids most common?
|
quarter horses
|
|
|
what are the four types of sarcoids?
|
1. verrucous
2. fibroblastic 3. mixed 4. occult |
|
|
how are sarcoids transmitted?
|
flies
|
|
|
why should you NEVER biopsy a sarcoid?
|
- treatment is ineffective
- it can convert to the more aggressive fibroblastic sarcoid |
|
|
in what circumstances is equine adenovirus transmitted to foals?
|
Equine adenovirus is an opportunistic pathogen, so FPT, and PSCID foals are most are most at risk for infection
|
|
|
what breed of foals are most predisposed to equine adenovirus type I infections? What is the disease and mortality rate?
|
- Arabian foals
- bronchopneumonia with close to 100% mortality |
|
|
what are three clinical signs in FPT foals with an equine adenovirus infection?
|
1. Acute URT
2. conjunctivitis 3. bronchopneumonia |
|
|
what are two important histopathological indicators of equine adenovirus?
|
1. basophilic intranuclear inclusion bodies
2. bronchopneumonia |
|
|
to which genus does horse pox virus belong? What are the two major syndromes that it causes?
|
- Orthopox (note, that this is a notifiable disease)
1. contagious papular stomatitis (CPD in notes??) 2. grease heel |
|
|
what are the three important equine alphaherpesviruses and the diseases they cause?
|
1. EHV-1: equine abortion
2. EHV-3: coital exanthema 3. EHV-4: rhinopneumonitis (rarely abortion) |
|
|
what are the two most important equine gammaherpesviruses and the diseases they cause?
|
1. EHV-2: pharyngitis and URT infection
2. EHV-5: URT infection |
|
|
which equine herpesviruses cause abortion?
|
EHV-1 and EHV-4 (rare)
|
|
|
which equine herpesvirus causes coital exanthema?
|
EHV-3
|
|
|
which equine herpesvirus causes rhinopneumonitis and rarely, abortions?
|
EHV-4
|
|
|
which equine herpesvirus causes pharyngitis and URT?
|
EHV-2
|
|
|
which equine herpesviruses cause URT infection?
|
EHV-2 and EHV-5
|
|
|
EHV-1:
- how long does it shed? - in latent infections, where does it reside - what can reactivate infection? - describe the shedding process - why does the immune system have trouble getting rid of it? |
- sheds 15 days
- latent in CD8+ T-cells and the trigeminal ganglia - reactivated by stress - often reactivation causes silent reactivation (shedding with no clinical signs); nasal shedding - poor immunity due to evasion (close to 70 proteins) |
|
|
describe the life cycle of EHV-1 (i.e. the role of latency and abortion)
|
1. latently infected horse → infection reactivates
2. nasal shedding → causes pregnant mares to abort; also infection of young horses → 3. young horse reestablishes latency → the cycle continues |
|
|
what three organ systems are affected by EHV-1?
|
1. respiratory
2. repro 3. CNS |
|
|
EHV-1 abortions:
- which trimester? - how do abortions present clinically? - when are mares refractory to aborting? - what are come histopath findings in aborted tissue? - what are some gross pathologic findings? |
- 3rd trimester (> 180 days)
- abortions: intrauterine death and absence of autolysis - mares are refractory < 120 days - intranuclear inclusion bodies in lungs and liver - Jaundice, edema, enlarged spleen, hepatic necrosis |
|
|
what are the two EHV-1 syndromes that predominately affect the respiratory system?
|
1. respiratory form
2. pulmonary vasculotropic |
|
|
what are three clinical signs of the respiratory form of EHV-1 infection?
|
1. mild nasal discharge, coughing, dyspnea
2. young foals - pneumonitis 3. progressive lymphadenopathy |
|
|
what are four clinical signs of the pulmonary vasculotropic form of EHV-1 infection?
|
1. sudden onset
2. high fever 3. respiratory distress 4. multisystemic vasculitis |
|
|
what are two neurological forms of EHV-1 infection?
|
1. EHV-1 myeloencephalopathy
2. ocular form |
|
|
EHV-1 in newborn foals:
- when do they get sick? - prognosis - clinical signs - what causes death? |
- sick at birth or within 1-2 days
- poor prognosis - fail to nurse, lethargic, fever, leukopenia, hypoxic, severe respiratory distress - they die via viral pneumonia and respiratory failure |
|
|
what are the three syndromes associated with EHV-4 infection?
|
1. rhinopneumonitis
2. genital form 3. neurologic form (rare) |
|
|
EHV-4 rhinopneumonitis
- age most susceptible - most severe clinical sign |
- < 2 years
- acute bronchopneumonia |
|
|
comment on the outcome of the genital form of EHV-4 infection
|
abortions in high virulence strains
|
|
|
how is EHV controlled and prevented?
|
- management, hygiene, and vaccination
- robust, but transient immunity - vaccinate mares early in gestation |
|
|
comment on the pros and cons of EHV vaccination:
- type of immune response - degree of protection - risks |
- poor CMI response
- enhanced cytotoxic T-lymphocyte (CTL) response - not completely protective - may allow for silent transmission of the virus |
|
|
what acronym is used for EHV prevention?
|
- SISS
- Segregation - Isolation - Subdivision - Stress reduction |
|
|
what acronym is used for EHV outbreak control?
|
- DISH
- Disinfection - Isolation - Sampling - Hygiene |
|
|
what disease is caused by EHV-3?
|
coital exanthema
|
|
|
describe the progression of lesions associated with EHV-3 (coital exanthema)
|
papules → vesicles → pustules → erosive hemorrhagic ulcers on the vulva and penis
|
|
|
where does coital exanthema appear on foals?
|
on the muzzle and lips
|
|
|
what lesions does EHV-5 produce?
|
pulmonary fibrosis in adult horses
|
|
|
when should a mare be vaccinated for EHV to prevent abortion during pregnancy?
|
5, 7, and 9 months of gestation
|
|
|
describe influenza virus nomenclature
|
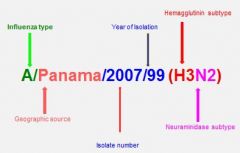
|
|
|
what are clinical signs of the respiratory-abortion form of equine viral arteritis
|
- fever
- edema - photophobia, corneal opacity - urticaria - abortions after 3 months (compare with EHV) - ataxia, dyspnea, diarrhea - fulminating interstitial pneumonia in foals |
|
|
which equine virus causes abortion after only 3 months?
|
equine viral arteritis
|
|
|
what are some gross lesions PM caused by equine viral arteritis?
|
- ***splenic infarcts
- ***necrotizing panvasculitis - edema of subcutis - fluid in cavities - pneumonia, enteritis - fetuses partially autolyzed, no vascular lesions |
|
|
what are five differentials for equine viral arteritis?
|
1. EIA (equine infectious anemia)
2. influenza 3. EHV-1 4. EHV-4 5. Getah |
|
|
if you have a stallion infected with equine viral arteritis, culling is the recommendation; however, if the owner insists on breeding, what is the proper protocol?
|
- test breed in two seronegative mares
- 28 days post-breeding, check for neutralizing antibodies to EAV - if mares are seronegative, stallion is qualified for breeding and classified as "non-shedding) - if mares seroconvert, the stallion is a shedder; needs to be approved by the State veterinarian for breeding; carrier should only be bred to mares that are seropositive by natural exposure or vaccination |
|
|
what type of viruses are EEE, WEE, and VEE?
|
Togaviridae
|
|
|
what type of viruses are JEV and WNV?
|
flaviviruses
|
|
|
which six types of viruses causes are equine encephalitides?
|
1. EEE
2. WEE 3. VEE 4. JEV 5. WNV 6. Hendravirus |
|
|
which animals are reservoir hosts for
- EEEV? - WEEV? - VEEV? |
- EEEV and WEEV: passerine birds
- VEEV: rodents |
|
|
how are most arboviral encephalitides transmitted?
|
via arthropod vectors (e.g. mosquitos)
|
|
|
which animals are the reservoir in the mosquito-reservoir cycle for:
- EEEV? - WEEV? - VEEV? |
- EEEV: bird-mosquito
- WEEV: bird-mosquito - VEEV: rodent-mosquito |
|
|
explain the biphasic pathogenesis of the equine encephalitis viruses
|
- primary viremia and replication in 2-5 days (pyrexia, depression and anorexia)
- virus in aerosol in urine - secondary CNS replication at 6-8 days with paralysis of the lips, drooping eyelids, and incoordination |
|
|
which specimens are collected to diagnose EEE/WEE/VEE?
|
serum, CSF, brain, spinal cord
|
|
|
how are EEE/WEE/VEE prevented and controlled?
|
- sentinel animals (e.g. rodents)
- quarantine - control insects - inactivated and MLV vaccines - VEEV: vaccinate border animals |
|
|
west nile fever is caused by what type of virus?
|
flavivirus
|
|
|
what are some clinical signs for west nile fever?
|
- muscle fasciculations
- mentation/behavior changes - lethargy, weakness, ataxia, paralysis |
|
|
what is the prognosis of a horse with west nile fever?
|
most recover
|
|
|
what animals are very susceptible, with a high mortality, to west nile virus?
|
crows (corvids)
|
|
|
what are three ways that WNV is diagnosed?
|
1. VI or serology
2. CSF 3. RT-PCR |
|
|
which animal is the reservoir in the mosquito-reservoir cycle for WNV?
|
birds such as ducks and geese
|
|
|
describe the clinical signs for the three phases of WNV in birds; comment on likelihood of recovery
|
- Phase I: depression, anorexia, sleepy; recovery likely
- Phase II: Phase I signs plus greenish diarrhea, central blindness, ataxia; recovery with intense therapy -Phase III: seizures, tremors, death |
|
|
what are four ways to control WNV?
|
1. mosquito control
2. equines: vaccination (booster at 3-6 weeks) 3. DNA vaccine 4. canarypox vectored vaccine |
|
|
equine infectious anemia is what type of virus?
|
Retroviridae (a Lentivirus)
|
|
|
what are four ways that equine infectious anemia is transmitted?
|
1. large biting flies
2. in utero 3. colostral 4. iatrogenic (most common) - virus survives in syringe for 4 days |
|
|
how long are horses carriers for EIA?
|
life
|
|
|
what disease does EIAV cause?
|
Swamp Fever
|
|
|
where does EIAV replicate?
|
in tissue macrophages, but not monocytes
|
|
|
how does EIAV cause anemia?
|
- ↓hematopoiesis
- complement mediated hemolysis |
|
|
describe the course of disease caused by EIAV
|
- it has a dynamic and defined course with recurrence
- recurring fever that coincides with titer |
|
|
how does EIAV affect the kidney?
|
virus-Ab complexes cause subclinical glomerulonephritis
|
|
|
what are three disease states of EIAV infection?
|
acute, chronic, and asymptomatic
|
|
|
what are clinical signs of acute EIAV infection?
|
- fever
- jaundice - subcutaneous edema - lethargy - petechiae - anemia - thrombocytopenia - sideroleukocytes |
|
|
what are clinical signs for chronic EIAV infection?
|
recurring fever (105 - 108 °F)
- anemia - edema |
|
|
foal gastroenteritis virus:
- family, genus, group - age affected - clinical signs |
- Reoviridae, Rotavirus, Group A
- foals < 2week - 3 months - watery, non-fetid diarrhea; enteritis; self-limiting infection |
|
|
how is foal gastroenteritis virus prevented and controlled?
|
- hygiene
- inactivated group A vaccine |
|
|
equine influenza virus
- family, group - genome - envelope? - what two strains have been found in horses? |
- Orthomyxoviridae, Influenza A virus
- (-) sense ssRNA, 8 gene segments - two envelope proteins: H and N - A/equi 1 (H7N7) and A/equi 2 (H3N8) |
|
|
Which species are susceptible to each type of influenza virus (A, B, and C)?
|
-A: humans, horses, swine, birds
-B: humans -C: humans and swine |
|
|
what group of rotaviruses infect horses?
|
Group A
|
|
|
which strain of virus was just isolated from bats?
|
H17N10
|
|
|
what is antigenic shift, with regards to influenza?
|
exchange of gene segments
|
|
|
what is antigenic drift, with regards to influenza?
|
mutation of the genome, thus coding for a new gene (compared to antigenic shift, which is exchange without mutation)
|
|
|
comment on the incubation period of equine influenza virus
|
short: < 2 days
|
|
|
what are some clinical signs of equine influenza?
|
- fever
- strong, dry cough - nasal discharge: serous to mucoid - pneumonia in foals - post-infection encephalopathy (foals) |
|
|
comment on the spreading of influenza by horses that have recovered from the disease
|
- they are MECHANICAL carriers of the disease, but they do not have a latent infection
|
|
|
comment on horses vaccinated for influenza, that acquire the virus
|
they can be infected and shed the virus for several days without showing overt clinical signs
|
|
|
why do horses with influenza cough?
|
because the respiratory cilia are destroyed
|
|
|
what are six differentials for equine influenza?
|
1. equine adenovirus 1
2. equine herpesvirus 1 3. equine herpesvirus 4 4. equine viral arteritis 5. Strep equi 6. Strep zooepidemicus |
|
|
what are the three types of vaccines available for equine influenza?
|
1. inactivated
2. MLV, cold adapted 3. canary pox vector |
|
|
which influenza vaccine is
- given for pre-foaling? - for foals > 4 months? - given as early as 1 month? - gives 12-month protection? |
- given for pre-foaling: inactivated
- for foals > 4 months: canary pox vectored - given as early as 1 month: inactivated - gives 12-month protection: MLV |
|
|
what are two circumstances where MLV equine influenza vaccine is contraindicated?
|
- foals < 11 months
- pregnant mares |
|
|
what are the gross pathologic lesions associated with acute EIAV?
|
lymphadenopathy, hepato-splenomegaly, anemia, hemorrhages, edema
|
|
|
what are the histopathologic lesions associated with acute EIAV?
|
hepatic lipidosis, hepatocellular necrosis, hemosiderosis, perivascular lymphocytic infiltration
|
|
|
how is EIAV prevented and controlled?
|
- test and slaughter
- quarantine (200 yard rule; tabanid files will only strike one horse if it is 200 yards away or more from other horses) - one horse, one needle, one syringe rule - clean and sterilize instruments - insect control - do not intermingle - quarantine new arrivals - do not breed reactors |
|
|
how are warts treated on horses?
|
cryosurgery, TFA, or podophyllin
|
|
|
what procedures are used to diagnose equine adenovirus?
|
VI, PCR and/or serology by HI/SN
|
|
|
what procedures are used to diagnose equine herpesvirus?
|
- blood in EDTA and nasal swab for PCR/real-time PCR
- Blood in EDTA and nasal swab for VI if clinical signs and PCR are positive - paired sera samples for antibodies - differential ELISA for EHV 1/4 - CSF in EHM - viral antigen detection (IHC, in situ hybridization, and PCR) |
|
|
what procedures are used to diagnose equine viral arteritis?
|
- VI from secretions and fluids
- RT-PCR (citrated whole blood, semen, nasopharyngeal swabs, fetus) - serology: VN is the official text (1:4 titer or greater is positive) - test mating |
|
|
what tests are used to diagnose equine encephalitis viruses?
|
- IgM capture ELISA for antibodies
- real-time PCR - viral antigen by IFA in cryostat sections of brain - submit to a lab for virus isolation where they will culture in a rodent or bird egg |
|
|
what tests are used to diagnose west nile virus?
|
- VI or serology (IgM antigen capture ELISA)
- CSF - RT-PCR |
|
|
what tests are used to diagnose foal gastroenteritis virus?
|
- VI
- PAGE for dsDNA (polyacrylamide gel electrophoresis) - Immuno-EM |
|
|
what lab tests are used to diagnose foal gastroenteritis virus?
|
- VI from nasal swabs
- RT-PCR - Serology: HI, single radical hemolysis |
|
|
what tests are used to definitely diagnose foal gastroenteritis virus?
|
- VI by EM or Immuno-EM
- PAGE for ds-RNA |
|
|
what tests are used to diagnose equine infectious anemia virus?
|
- Coggin's test**
- VI - PCR for provirus DNA - precipitating antibodies: 2-4 weeks - neutralizing antibodies: 40 days - antibody diagnostic tests (AGID, ELISA) |
|
|
what is the international gold standard to diagnose equine infectious anemia virus?
|
Coggin's test
|
|
|
which horse viruses are reportable?
|
Horse Pox, EHV, EEE, WEE, VEE, west nile, EIA
|
|
|
if an equine papilloma does not regress by 6 months, what should you suspect?
|
immunodeficiency
|
|
|
why is biopsy of a verrucous, static, or occult sarcoid not indicated?
|
because it may lead to the aggressive fibrous type
|
|
|
comment on the survivability of equine adenovirus in the environment.
|
the virus is non-enveloped and may survive for a long time in the environment
|
|
|
why is horse pox reportable?
|
because it must be distinguished from vesicular stomatitis
|
|
|
why are herpesviruses so successful (besides their charming personalities)?
|
because they have extended periods of latency and recrudesce to infect naïve horses
|
|
|
how do you differentiate EHV-1 from EHV-4?
|
- type-specific PCR
- Glycoprotein G ELISA |
|
|
how does fever differ between WEE/EEE and VEE?
|
EEE/WEE are biphasic, whereas VEE is rarely biphasic
|
|
|
why is heparinized blood not suitable for definitive ID of equine viral arteritis?
|
because heparin interferes with virus isolation
|
|
|
what is the most specific test for EIAV? How does this fit into the testing regimen?
|
AGID is more specific, but less sensitive. Therefore, it is used as a confirmatory test rather than a screening test.
|

