![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
45 Cards in this Set
- Front
- Back
|
GLY
|
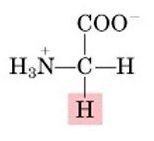
Glycine, G
Nonpolar, aliphatic |
|
|
ALA
|
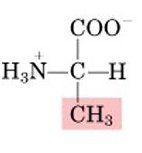
Alanine, A
Nonpolar, aliphatic |
|
|
VAL
|
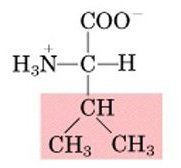
Valine, V
Nonpolar, aliphatic |
|
|
LEU
|
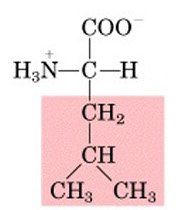
Leucine, L
Nonpolar, aliphatic |
|
|
MET
|
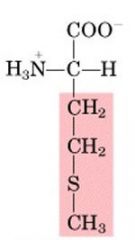
Methionine, M
Nonpolar, aliphatic |
|
|
ILE
|
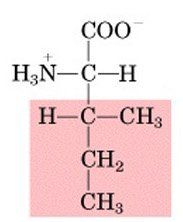
Isoleucine, I
Nonpolar, aliphatic |
|
|
PHE
|
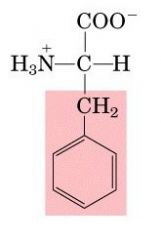
Phenylalanine, F
Aromatic |
|
|
TYR
|
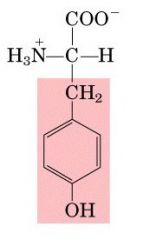
Tyrosine, Y
Aromatic, polar side group |
|
|
TRP
|
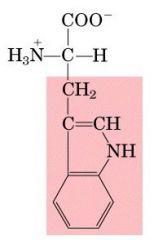
Tryptophan, W
Aromatic |
|
|
SER
|
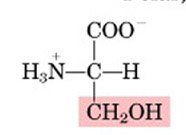
Serine, S
Polar, uncharged |
|
|
THR
|
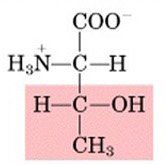
Threonine, T
Polar, uncharged |
|
|
CYS
|
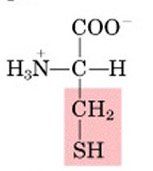
Cysteine, C
Polar, uncharged |
|
|
PRO
|
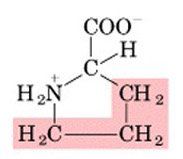
Proline, P
Polar, uncharged |
|
|
ASN
|
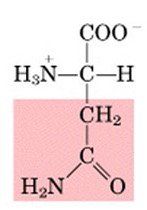
Asparagine, N
Polar, uncharged |
|
|
GLN
|
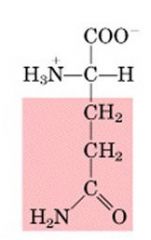
Glutamine, Q
Polar, uncharged |
|
|
ASP
|
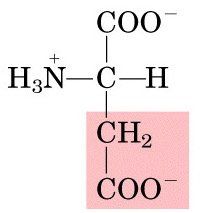
Aspartic Acid, D
Polar, negatively charged |
|
|
GLU
|
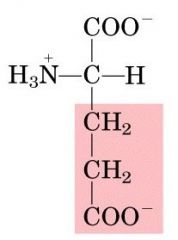
Glutamic Acid, E
Polar, negatively charged |
|
|
LYS
|
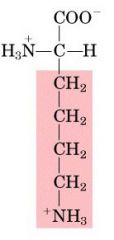
Lysine, K
Polar, positively charged |
|
|
ARG
|
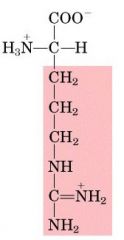
Arginine, R
Polar, positively charged |
|
|
HIS
|
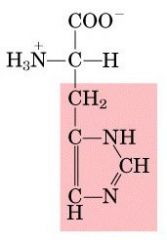
Histidine, H
Polar, positively charged |
|
|
Three-dimensional structure of amino acids
|
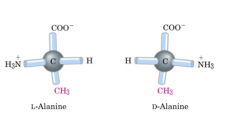
The amino acids are all chiral with the exception of glycine, whose side chain is H. As with lipids, biochemists use the L and D nomenclature
|
|
|
Eg: Stereochemistry of Alanine (Fischer Proj)
|
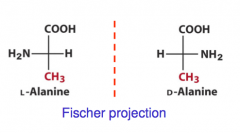
|
|
|
Eg: Stereochemistry of Alanine (Perspective View)
|
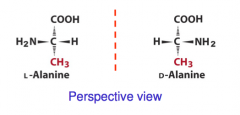
|
|
|
α-Amino acids: Properties
|
1. Crystalline solids which melt with decomposition at fairly high temperatures.
2. Insoluble in nonpolar solvents and appreciably soluble in water |
|
|
Explanation of properties
|
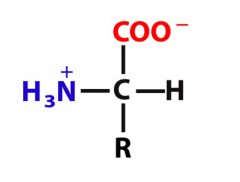
Intramolecular acid–base reaction: the H from the COOH group is transferred to the –NH2 group.
A neutral dipolar ion, an ion that has one (+) charge and one (-) charge, forms. Neutral dipolar ions are known as zwitterions. |
|
|
Zwitterions or dipolar ions
|
A zwitterion (dipolar ion) is a compound with no overall electrical charge, but contains separate parts which are positively and negatively charged.
|
|
|
Zwitterions or dipolar ions (cont)
|
Amino acids exist in this form even in the solid state
--> Ionic attractions form between the molecules --> High melting points. |
|
|
Amino acids are amphiprotic
|
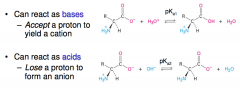
|
|
|
Acidity: α-COOH groups
|
The average pKa of an α-carboxyl group is 2.19, which makes them considerably stronger acids than acetic acid (pKa 4.78).
The greater acidity is accounted for by the electron- withdrawing inductive effect of the adjacent –NH2 group. |
|
|
Basicity: α-NH2 groups
|
The average value of pKb for an α-NH3+ group is 9.47, which
makes them less basic than a primary alkyl ammonium ion. |
|
|
Acidity: side chain -COOH
|
Due to the electron-withdrawing inductive effect of the α- NH2 group, side chain -COOH groups are also stronger than acetic acid.
– The effect decreases with distance from the α-NH2 group. Compare: α-COOH group of alanine (pKa 2.35) β-COOH group of aspartic acid (pKa 3.86) γ-COOH group of glutamic acid (pKa 4.07) |
|
|
Acid-base properties of amino acids
|
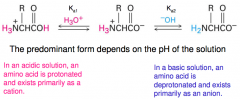
In aqueous solution an equilibrium exists between the dipolar ion, the cationic and the anionic forms of the amino acid
|
|
|
Isoelectric point
|
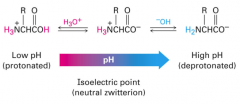
At some intermediate pH called the pI (isoelectric point), the concentration of the dipolar ion is at a maximum and the concentrations of anionic and cationic forms are equal. At this pH the net charge is zero; amino acid is least soluble in water and does not migrate in electric field
|
|
|
Isoelectric point: For amino acid without ionizable side chains, the isoelectric point (equivalence point, pI) is
|
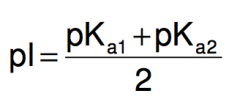
|
|
|
Each individual amino acid has a characteristic pI
|

|
|
|
pKa of amino acids I.
|
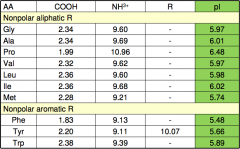
|
|
|
pKa of amino acids II
|
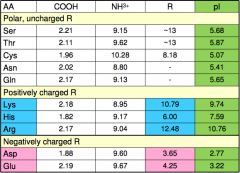
|
|
|
D-amino acids
D-amino acids are found in a few small peptides, including some peptides of bacterial cell walls and certain antibiotics (such as penicillin). |
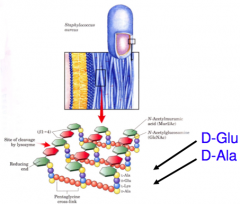
|
|
|
Amino acids with two chiral centers
|
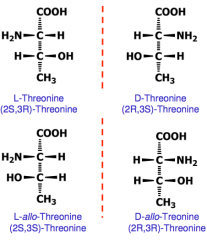
|
|
|
Chemical resolution of amino acids
|
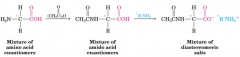
• The amino group is acetylated.
• Diastereomers are formed with a base (e.g. brucine). • Diastereomers are separated and the enantiomers are regenerated from them. |
|
|
Enzymatic resolution
|
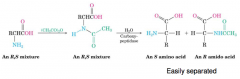
Enzymes selectively catalyze the hydrolysis of an acetyl group from the amino group of an L-amino acid.
|
|
|
Synthesis of amino acids I
|
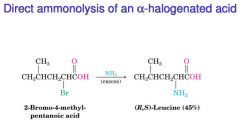
|
|
|
Synthesis of amino acids II
|
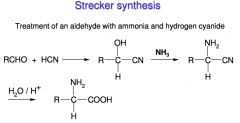
|
|
|
Reactions of amino acids
|

|
|
|
Reactions of amino group
|
1. Saltformation
2. Alkylation 3. Arylation 4. Acylation 5. Reaction w/ nitrous acid 6. Schiff’s base formation 7. Reaction w/ ninhydrin 8. Transamination |

