![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
50 Cards in this Set
- Front
- Back
|
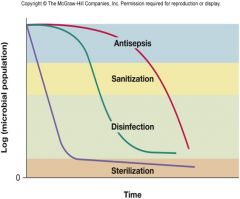
sterilization
|
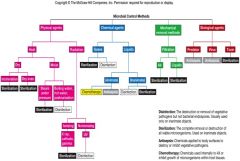
destruction or removal of all viable organisms
|
|
|
disinfection
|
killing, inhibition, or removal of disease causing (pathogenic) organisms
|
|
|
disinfectants
|
agents, usually chemical, used for disinfection
usually used on inanimate objects |
|
|
sanitization
|
reduction of microbial population to levels deemed safe (based on public health standards)
|
|
|
antisepsis
|
Destruction or inhibition of microorganisms on living tissue
|
|
|
antiseptics
|
chemical agents that kill or inhibit growth of microorganisms when applied to tissue
|
|
|
chemotherapy
|
use of chemicals to kill or inhibit growth of microorganisms within host tissue
|
|
|
Antimicrobial Agents
|
agents that kill microorganisms or inhibit their growth
cidal agents kill static agents inhibit growth |
|
|
-cidal Agents
|
suffix indicating that agent kills
|
|
|
germicide
|
kills pathogens and many nonpathogens but not necessarily endospores
|
|
|
-static Agents
|
suffix indicating that agent inhibits growth
include bacteriostatic and fungistatic |
|
|
The Pattern of Microbial Death
|
microorganisms are not killed instantly
population death usually occurs exponentially measure of agent’s killing efficiency |
|
|
decimal reduction time (D-value)
|
time to kill 90% of microorganisms and spores in a sample under specific conditions
|
|
|
Z- value
|
temperature change needed to kill 90% of microorganisms and spores in a sample under specific conditions
|
|
|
persister cells
|
must be sure persister cells (viable but nonculturable (VBNC)) are dead
once they recover they may regain the ability to reproduce and cause infection |
|
|
Conditions Influencing the Effectiveness of Antimicrobial Agent Activity
|
larger populations take longer to kill than smaller populations
|
|
|
population composition
|
microorganisms differ markedly in their sensitivity to antimicrobial agents
|
|
|
concentration or intensity of an antimicrobial agent
|
usually higher concentrations or intensities kill more rapidly (not always, e.g., EtOH)
relationship is not linear |
|
|
Contact time
|
duration of exposure
longer exposure = more organisms killed |
|
|
temperature
|
higher temperatures usually increase amount of killing
|
|
|
local environment
|
many factors (e.g., pH, viscosity, and concentration of organic matter) can profoundly impact effectiveness
organisms in biofilms are physiologically altered and less susceptible to many antimicrobial agents |
|
|
Moist Heat
|
destroys viruses, fungi, and bacteria
boiling will not destroy spores and does not sterilize degrades nucleic acids, denatures proteins, and disrupts membranes |
|
|
Steam Sterilization
|
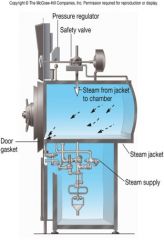
must be carried out above 100oC which requires saturated steam under pressure
carried out using an autoclave (Developed by Chamberland, 1884) (121 C at 15 psi for 10-15 min) effective against all types of microorganisms including spores quality control - includes strips with Geobacillus stearothermophilus |
|
|
Pasteurization
|
controlled heating at temperatures well below boiling (55 - 60 C)
used for milk, beer, and other beverages process does not sterilize but does kill pathogens present and slows spoilage by reducing the total load of organisms present Introduced to US in 1889 |
|
|
Tyndallization
|
Developed by John Tyndall
Intermittent sterilization 30-60 min of steam repeated 3 times with 23-24 hr incubations between Used to kill spores – spores germinate and are then killed by the steam treatment |
|
|
Dry Heat Sterilization
|
less effective than moist heat sterilization, requiring higher temperatures and longer exposure times (e.g. incineration or oven)
items subjected to 160–170oC for 2 to 3 hours oxidizes cell constituents and denatures proteins E.g., C. botulium – 5 min at 121C or 2 hrs at 160C |
|
|
Dry Heat Incineration
|
bench top incinerators are used to sterilize inoculating loops used in microbiology laboratories
|
|
|
Filtration
|
reduces microbial population or sterilizes solutions of heat-sensitive materials by removing microorganisms
also used to reduce microbial populations in air Can use depth filters or membrane filters |
|
|
membrane filters
|
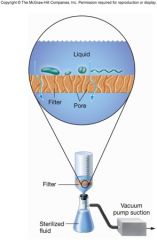
-porous membranes with defined pore sizes that remove microorganisms primarily by physical screening
-have replaced depth filters for the most part -Removes microorganisms from heat sensitive liquids (e.g., pharmaceuticals, antibiotics, vitamin solutions, culture media, etc) |
|
|
high-efficiency particulate air (HEPA) filters
|
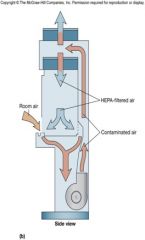
used in laminar flow biological safety cabinets
Removes 99.97% of particles 0.3 um or larger |
|
|
Ultraviolet (UV) Radiation
|
-wavelength of 260 is most bactericidal (DNA absorbs)
-causes thymine dimers in DNA preventing replication and transcription -UV limited to surface sterilization because it does not penetrate glass, dirt films, water, and other substances -has been used for water treatment |
|
|
Ionizing Radiation
|
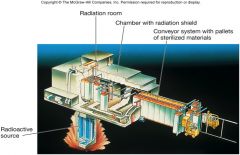
-gamma radiation penetrates deep into objects
-destroys bacterial endospores; not always effective against viruses -used for sterilization and pasteurization of antibiotics, hormones, sutures, plastic disposable supplies, and food |
|
|
Chemical Agents
|
-A disinfectant must be effective against a wide variety of infectious agents at low concentrations
-must be effective in the presence of organic matter; should be stable in storage -Pleasant odor, soluble in water and lipids, low surface tension and relatively inexpensive -overuse of antiseptics such as triclosan has selected for triclosan resistant bacteria and possibly antibiotic resistance (e.g., Pseudomonas aeruginosa) |
|
|
Phenolics
|
Phenol was 1st widely used antiseptic and disinfectant. Used in 1867 by Joseph Lister
Commonly used as laboratory and hospital disinfectants (e.g., Lysol) Act by denaturing proteins and disrupting cell membranes Tuberculocidal, effective in presence of organic material, and long lasting Disagreeable odor and can cause skin irritation E.g., triclosan is used in hand sanitizers |
|
|
Alcohols
|
-among the most widely used disinfectants and antiseptics
-two most common are ethanol and isopropanol (60 – 80 %) -bactericidal, fungicidal, but not sporicidal -inactivate some viruses -denature proteins and possibly dissolve membrane lipids |
|
|
Halogens
|
any of five elements: fluorine, chlorine, bromine, iodine, and astatine
important antimicrobial agents |
|
|
Halogens - Iodine
|
-skin antiseptic
-oxidizes cell constituents and iodinates proteins -at high concentrations may kill spores -skin damage, staining, and allergies can be a problem -E.g., Wescodyne |
|
|
iodophore
|
iodine complexed with organic carrier
released slowly to minimize skin burns |
|
|
Halogens - Chlorine
|
-oxidizes cell constituents
-important in disinfection of water supplies and swimming pools, used in dairy and food industries, -effective household disinfectant -destroys vegetative bacteria and fungi, -chlorine gas is sporicidal -can react with organic matter to form carcinogenic compounds -E.g., Bleach is highly effective (10% with a little vinegar) |
|
|
Heavy Metals
|
-e.g., ions of mercury, silver, arsenic, zinc, and copper (e.g., CuSO4 as algaecide in swimming pools, Silver nitrate to prevent ophthalmic gonorrhea)
-effective but usually toxic -combine with and inactivate proteins; may also precipitate proteins |
|
|
Quaternary Ammonium Compounds
|
-detergents that have antimicrobial activity and are effective disinfectants
-amphipathic organic cleansing agents -act as wetting agents and emulsifiers -Disrupt membranes and denature proteins -kill most bacteria, but not Mycobacterium tuberculosis or endospores -safe and easy to use, but inactivated by hard water and soap -E.g., benzalkonium chloride (food utensils) |
|
|
Aldehydes
|
-commonly used agents are formaldehyde and glutaraldehyde
-highly reactive molecules -sporicidal and can be used as chemical sterilants -combine with and inactivate nucleic acids and proteins |
|
|
Chemical Methods of Microbial Control
|

Disk-diffusion method
|
|
|
Sterilizing Gases
|
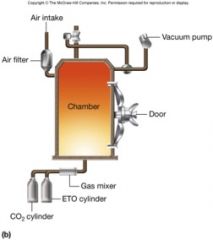
-used to sterilize heat-sensitive materials like plastics
-microbiocidal and sporicidal -ethylene oxide sterilization is carried out in equipment resembling an autoclave - rapidly penetrates packing materials, even plastic wraps (5-8 hrs at 38C or 3-4 hrs at 54C at 40-50% RH) – very toxic to humans -betapropiolactone and vaporized hydrogen peroxide -combine with and inactivate DNA and proteins |
|
|
Evaluation of Antimicrobial Agent Effectiveness
|
-complex process regulated by U.S. federal agencies:
Environmental Protection Agency-regulates disinfectants Food and Drug Administration-regulates agents used on humans or animals |
|
|
Phenol Coefficient Test
|
-Best known disinfectant screening test
-potency of a disinfectant is compared to that of phenol -useful for screening but may be misleading -Uses carefully controlled conditions that don’t really resemble actual use conditions -Uses standard amounts of Salmonella enterica serovar Typhi and Staphylococcus aureus – looks for the highest dilution that kills bacteria in 10 min. |
|
|
Other Evaluation Methods-dilution test
|
determines rate at which selected bacteria are destroyed by various chemical agents by treating contaminated stainless steel carriers
|
|
|
Other Evaluation Methods-normal in-use testing
|
testing done using conditions that approximate normal use of disinfectant
|
|
|
Biological Control of Microorganisms
|
emerging field showing great promise – using microorganisms as natural predators
natural control mechanisms |
|
|
natural control mechanisms
|
-predation by Bdellovibrio
-viral-mediated lysis using pathogen specific bacteriophage lysins -toxin-mediated killing using bacteriocins |

