![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
18 Cards in this Set
- Front
- Back
|
Chemical nomenclature and creating chemical formulas: Ionic Compounds |
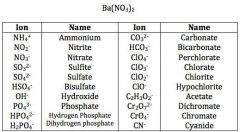
|
|
|
Look in book for polyatomic ions and compounds to study |
Yay |
|
|
Word and balanced skeleton equations |
Example: 2Na+Cl2= 2NaCl Look in the chemistry book to study for the rest |
|
|
Types of Chemical Reactions |
1. Synthesis Example: sodium + oxygen= sodium oxide 2. Decomposition Example: Magnesium oxide= magnesium + oxygen 3. Single Displacement Example: Chlorine + potassium bromide= potassium chloride + bromine 4. Double Displacement Example: barium chloride+ potassium iodide= barium iodide + potassium chloride 5. Combustion Example: methane + oxygen= copper + water 6. Neutralization Example: NaOH + HCl = NaCl + H2O |
|
|
Combustion of hydrocarbons |
Type of reaction where where a hydrocarbon reacts with oxygen to create Carbon Dioxide, water and heat. Also they are molecules with hydrogen and carbon. |
|
|
Rates of Chemical Reactions |
1. Tempature Example: increasing tempature is going to increase the rate of reaction because of increase in high energy collisions. 2. Concentration Example: If the concentration of a dissolved reactant is increased, the reactant particles become more crowded. 3. Surface Area Example: If the surface area if a reactant is increased, then the rate of reaction will be raised. 4. Catalysts Example: Catalysts raise the rate if reaction without being used up,they do this by powering the energy activation needed. |
|
|
The PH Scale, Indicators and explanation of the scale |
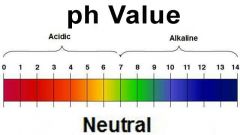
A PH Indicator is a chemical that is added in small amounts to a solution to visually show the audicity or basicity of the solution by changing colours within a small range of PH values. A PH Scale ranges from 0-14 and is used to classify aqueous solutions as acidic, basic or neutral. |
|
|
Acids and Bases and Neutralization |
Acids are compounds that have a spur taste, when dissolved in water, they produce Hydrogen Ions. They also have binary and oxo acids. Bases are compounds that have a bitter taste. Many bases are ionic compounds that separate into metal ions and hydroxide ions. Neutralization: when a compound is neither acidic or alkaline and has a PH of 7. |
|
|
Periodic Table |

|
|
|
Chemical and Physical properties of matter, including metals, non-metals, metalloids, halogens, noble gases |
Physical Properties: Metal:metals differ widely in hardness, ductility, maellebility, density and melting point. Non-metal: most non metals sit at room tempature . They can be very brittle and are poor conductors of electricity and heatMetalloids: typically solids and have a metallic luster. Brittle electricity and has a average transmission of heat. Metalloids: typically solids and have a metallic luster. Brittle electricity and has a average transmission of heat. Halogens: have a low melting and boiling points and they become darker as your turn down the group of halogens. Noble Gases: order less and colour less and are Minatomic gases with a very low Chemical reaction. Chemical Properties: Metals: metals are very reactive,metal tends to lose electrons easy and form positive ions. Non-metal: tend to have a tendacy ti want to share electrons with other atoms. Metalloids: can form readly gases and they react with the halogens so they can form compounds. Noble gases: have a very low Chemical reactivity. |
|
|
Indicators of a chemical and physical change |
Physical Change: - a change in colour - a change in tempature - precipitation is formed - formation of a gas Chemical Change: - Bubbles or gas appears - a oddour forms - changes in the melting and boiling points |
|
|
Catalyst |
A catalyst is a substance that speeds up a chemical reaction, but is not consumed by a chemical reaction. |
|
|
Ionic, covalent and molecular bonds |
Look in book for more info. |
|
|
Specific Heat |
The heat required to raise the tempature of the unit mass of a given substance by a given amount |
|
|
Valence level |
Valence levels are levels of a bohr Rutherford diagram. In the first shell, it can go up to two electrons, but the rest can go to 8 electrons. |
|
|
Ions |
an atom or molecule with a net electric charge due to the loss or gain of one or more electrons. |
|
|
Bohr rutherford diagrams for atoms |
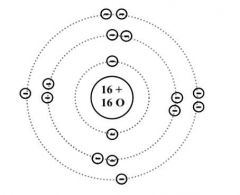
Look in book for more infor |
|
|
Bohr rutherford diagrams for ions |
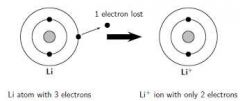
Look in book for more info |

